Abstract
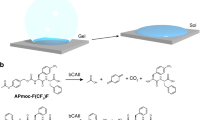
Drug-dependent dissociation or association of cellular receptors represents a potent pharmacologic mode of action for regulating cell fate and function1,2. Transferring the knowledge of pharmacologically triggered protein–protein interactions to materials science will enable novel design concepts for stimuli-sensing smart hydrogels. Here, we show the design and validation of an antibiotic-sensing hydrogel for the trigger-inducible release of human vascular endothelial growth factor3. Genetically engineered bacterial gyrase subunit B (GyrB) (ref. 4) coupled to polyacrylamide was dimerized by the addition of the aminocoumarin antibiotic coumermycin, resulting in hydrogel formation. Addition of increasing concentrations of clinically validated novobiocin (Albamycin) dissociated the GyrB subunits, thereby resulting in dissociation of the hydrogel and dose- and time-dependent liberation of the entrapped protein pharmaceutical VEGF121 for triggering proliferation of human umbilical vein endothelial cells. Pharmacologically controlled hydrogels have the potential to fulfil the promises of stimuli-sensing materials5,6,7,8,9 as smart devices for spatiotemporally controlled delivery of drugs within the patient.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Farrar, M. A., Alberol, I. & Perlmutter, R. M. Activation of the Raf-1 kinase cascade by coumermycin-induced dimerization. Nature 383, 178–181 (1996).
Hacker, H. et al. Specificity in Toll-like receptor signalling through distinct effector functions of TRAF3 and TRAF6. Nature 439, 204–207 (2006).
Ehrbar, M. et al. Cell-demanded liberation of VEGF121 from fibrin implants induces local and controlled blood vessel growth. Circ. Res. 94, 1124–1132 (2004).
Denhardt, D. T. DNA gyrase and DNA unwinding. Nature 280, 196–198 (1979).
Ehrick, J. D. et al. Genetically engineered protein in hydrogels tailors stimuli-responsive characteristics. Nature Mater. 4, 298–302 (2005).
Lendlein, A., Jiang, H., Junger, O. & Langer, R. Light-induced shape-memory polymers. Nature 434, 879–882 (2005).
Miyata, T., Asami, N. & Uragami, T. A reversibly antigen-responsive hydrogel. Nature 399, 766–769 (1999).
Wang, C., Stewart, R. J. & Kopecek, J. Hybrid hydrogels assembled from synthetic polymers and coiled-coil protein domains. Nature 397, 417–420 (1999).
Thornton, P. D., McConnell, G. & Ulijn, R. V. Enzyme responsive polymer hydrogel beads. Chem. Commun. (Camb) 47, 5913–5915 (2005).
Lutolf, M. P. et al. Repair of bone defects using synthetic mimetics of collagenous extracellular matrices. Nature Biotechnol. 21, 513–518 (2003).
Um, S. H. et al. Enzyme-catalysed assembly of DNA hydrogel. Nature Mater. 5, 797–801 (2006).
Toledano, S., Williams, R. J., Jayawarna, V. & Ulijn, R. V. Enzyme-triggered self-assembly of peptide hydrogels via reversed hydrolysis. J. Am. Chem. Soc. 128, 1070–1071 (2006).
Murakami, Y. & Maeda, M. DNA-responsive hydrogels that can shrink or swell. Biomacromolecules 6, 2927–2929 (2005).
Kopecek, J. Smart and genetically engineered biomaterials and drug delivery systems. Eur. J. Pharm. Sci. 20, 1–16 (2003).
Lutolf, M. P. & Hubbell, J. A. Synthetic biomaterials as instructive extracellular microenvironments for morphogenesis in tissue engineering. Nature Biotechnol. 23, 47–55 (2005).
Beebe, D. J. et al. Functional hydrogel structures for autonomous flow control inside microfluidic channels. Nature 404, 588–590 (2000).
Clardy, J., Fischbach, M. A. & Walsh, C. T. New antibiotics from bacterial natural products. Nature Biotechnol. 24, 1541–1550 (2006).
Gormley, N. A., Orphanides, G., Meyer, A., Cullis, P. M. & Maxwell, A. The interaction of coumarin antibiotics with fragments of DNA gyrase B protein. Biochemistry 35, 5083–5092 (1996).
Winter, H. H. & Chambon, F. Analysis of linear viscoelasticity of a crosslinking polymer at the gel point. J. Rheol. 30, 367–382 (1986).
Lutolf, M. P. & Hubbell, J. A. Synthesis and physicochemical characterization of end-linked poly(ethylene glycol)-co-peptide hydrogels formed by Michael-type addition. Biomacromolecules 4, 713–722 (2003).
Ehrbar, M., Metters, A., Zammaretti, P., Hubbell, J. A. & Zisch, A. H. Endothelial cell proliferation and progenitor maturation by fibrin-bound VEGF variants with differential susceptibilities to local cellular activity. J. Control Release 101, 93–109 (2005).
Eder, J. P., Wheeler, C. A., Teicher, B. A. & Schnipper, L. E. A phase I clinical trial of novobiocin, a modulator of alkylating agent cytotoxicity. Cancer Res. 51, 510–513 (1991).
Murren, J. R. et al. Phase I and pharmacokinetic study of novobiocin in combination with VP-16 in patients with refractory malignancies. Cancer J. 6, 256–265 (2000).
Aggarwal, S. What’s fueling the biotech engine? Nature Biotechnol. 25, 1097–1104 (2007).
Acknowledgements
We are grateful to M. Lütolf for his critical comments on the manuscript as well as his advice on the rheology measurements. We thank B. Keller for cloning plasmid pWW873, R. Wirz for infrared spectra analysis, S. Gobaa for expertise in rheology measurements, M. Rimann for providing HUVECs and J. Loebus for technical assistance. The project has been made possible owing to start-up financing by the GEBERT RÜF STIFTUNG (Grant No. GRS-042/07) to W.W.
Author information
Authors and Affiliations
Contributions
The experiments were designed by M.E., R.S. and W.W. and carried out by M.E., R.S., E.H.C. and W.W. The manuscript was written by M.F. and W.W. The principal investigator is W.W.
Corresponding author
Ethics declarations
Competing interests
ETH Zurich has filed an application for a patent on the technology described in this manuscript, of which M.F. And W.W. are inventors.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2157 kb)
Rights and permissions
About this article
Cite this article
Ehrbar, M., Schoenmakers, R., Christen, E. et al. Drug-sensing hydrogels for the inducible release of biopharmaceuticals. Nature Mater 7, 800–804 (2008). https://doi.org/10.1038/nmat2250
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat2250
This article is cited by
-
Hydrogel interfaces for merging humans and machines
Nature Reviews Materials (2022)
-
External triggering and triggered targeting strategies for drug delivery
Nature Reviews Materials (2017)
-
Autonomy and Fear of Synthetic Biology: How Can Patients’ Autonomy Be Enhanced in the Field of Synthetic Biology? A Qualitative Study with Stable Patients
Science and Engineering Ethics (2017)
-
Self-assembled hydrogels utilizing polymer–nanoparticle interactions
Nature Communications (2015)