Abstract
Purpose
Soil microbial biomass (SMB), as the source and sink of soil nutrients, and its stoichiometry play a key role in soil organic carbon (SOC) and nitrogen (N) mineralization. The objective of this study was to investigate the responses of SOC and N mineralization to changes in microbial biomass and SOC, N, and phosphorus (P) stoichiometry resulted from long-term fertilization regimes.
Materials and methods
Soil was sampled from a rice-wheat rotation system subjected to 37 years of nine fertilization treatments with different nutrient input amounts: control (CK), N alone, N combined with mineral phosphorus (NP), NP plus potassium (NPK), manure alone (M), and M combined with N (MN), NP (MNP), NPK (MNPK), and a higher rate of M with NPK (hMNPK). The sampled soil was incubated for the determination of SOC and N mineralization, C, N, and P stoichiometry of soil and SMB, and associated soil enzymes related to C and N cycling.
Results and discussion
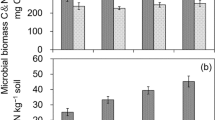
Relative to the CK and treatments with mineral fertilizers, treatments with manure (M, MN, MNP, MNPK, and hMNPK) significantly increased SOC and N mineralization by 48–78% and 54–97%, respectively. Microbial metabolic quotient (qCO2) decreased by 32–55% in treatments with manure compared to the N and NP treatments, but showed no effect on the qCO2 when compared to the NPK treatment. The leucine amino peptidase (LAP) enzyme showed significant positive correlation with SOC and N mineralization, and negatively related to the qCO2. Significantly negative correlations were also observed between SOC and N mineralization and soil C:P and N:P ratio, as well as microbial biomass SMBC:SMBP and SMBN:SMBP stoichiometry, respectively. However, the availability of N and P had limited effects on the qCO2 after reaching a certain value (0.69–0.72 mg CO2-C g−1 MBC h−1).
Conclusions
Lower soil elemental (C:P and N:P) and microbial biomass stoichiometry (SMBC:SMBP and SMBN:SMBP) and increase of LAP resulted from combined application of manure and mineral fertilizers, accelerated SOC, and N mineralization. Mineral nutrient input with manure amendments could be an optimal strategy to meet the microbial stoichiometric demands and enhance nutrient availability for crops in agricultural ecosystems.





Similar content being viewed by others
References
Aira M, Domínguez J (2014) Changes in nutrient pools, microbial biomass and microbial activity in soils after transit through the gut of three endogeic earthworm species of the genus Postandrilus Qui and Bouché. J Soils Sediments 1998:1335–1340. https://doi.org/10.1007/s11368-014-0889-1
Allison SD, Wallenstein MD, Bradford MA (2010) Soil carbon response to warming dependent on microbial physiology. Nat Geosci 3:336–340. https://doi.org/10.1038/ngeo846
Aziz T, Ahmed I, Farooq M, Maqsood MA, Sabir M (2011) Variation in phosphorus efficiency among Brassica cultivars I: international utilization and phosphorus remobilization. J Plant Nutr 34:2006–2017. https://doi.org/10.1080/01904167.2011.610487
Bowles TM, Acosta-Martínez V, Calderón F, Jackson LE (2014) Soil enzyme activities, microbial communities, and carbon and nitrogen availability in organic agroecosystems across an intensively-managed agricultural landscape. Soil Biol Biochem 68:252–262. https://doi.org/10.1016/j.soilbio.2013.10.004
Boye K, Noël V, Tfaily MM, Bone SE, Williams KH, Bargar JR, Fendorf S (2017) Thermodynamically controlled preservation of organic carbon in floodplains. Nat Geosci 10:415–419. https://doi.org/10.1038/ngeo2940
Brookes PC, Powlson DS, Jenkinson DS (1982) Measurement of microbial biomass phosphorus in soil. Soil Biol Biochem 14:319–329. https://doi.org/10.1016/0038-0717(82)90001-3
Cai A, Feng W, Zhang W, Xu M (2016) Climate, soil texture, and soil types affect the contributions of fine-fraction-stabilized carbon to total soil organic carbon in different land uses across China. J Environ Manag 172:2–9. https://doi.org/10.1016/j.jenvman.2016.02.009
Caruso G (2010) Leucine aminopeptidase, β-glucosidase and alkaline phosphatase activity rates and their significance in nutrient cycles in some coastal Mediterranean sites. Mar Drugs 8:916–940. https://doi.org/10.3390/md8040916
Chakrabarti K, Sarkar B, Chakraborty A, Banik P, Bagchi DK (2000) Organic recycling for soil quality conservation in a sub-tropical plateau region. J Agron Crop Sci 184:137–142. https://doi.org/10.1046/j.1439-037x.2000.00352.x
Chang EH, Chung RS, Tsai YH (2007) Effect of different application rates of organic fertilizer on soil enzyme activity and microbial population. Soil Sci Plant Nutr 53:132–140. https://doi.org/10.1111/j.1747-0765.2007.00122.x
Chen J, Chen D, Xu Q, Fuhrmann JJ, Li L, Pan G, Li Y, Qin H, Liang C, Sun X (2019) Organic carbon quality, composition of main microbial groups, enzyme activities, and temperature sensitivity of soil respiration of an acid paddy soil treated with biochar. Biol Fertil Soils 55:185–197. https://doi.org/10.1007/s00374-018-1333-2
Cleveland CC, Liptzin D (2007) C:N:P stoichiometry in soil: is there a “Redfield ratio” for the microbial biomass? Biogeochemistry 85:235–252. https://doi.org/10.1007/s10533-007-9132-0
Cui J, Zhu Z, Xu X, Liu S, Jones DL, Kuzyakov Y, Shibistova O, Wu J, Ge T (2020) Carbon and nitrogen recycling from microbial necromass to cope with C:N stoichiometric imbalance by priming. Soil Biol Biochem 142:107720. https://doi.org/10.1016/J.SOILBIO.2020.107720
DeForest JL (2009) The influence of time, storage temperature, and substrate age on potential soil enzyme activity in acidic forest soils using MUB-linked substrates and L-DOPA. Soil Biol Biochem 41:1180–1186. https://doi.org/10.1016/j.soilbio.2009.02.029
Dunn C, Jones TG, Girard A, Freeman C (2014) Methodologies for extracellular enzyme assays from wetland soils. Wetlands 34:9–17. https://doi.org/10.1007/s13157-013-0475-0
Fließbach A, Oberholzer H-R, Gunst L, Mäder P (2007) Soil organic matter and biological soil quality indicators after 21 years of organic and conventional farming. Agric Ecosyst Environ 118:273–284. https://doi.org/10.1016/J.AGEE.2006.05.022
Ge T, Yuan H, Zhu H, Wu X, Nie S, Liu C, Tong C, Wu J, Brookes P (2012) Biological carbon assimilation and dynamics in a flooded rice soil system. Soil Biol Biochem 48:39–46. https://doi.org/10.1016/j.soilbio.2012.01.009
Geisseler D, Horwath WR, Georg R, Ludwig B (2010) Pathways of nitrogen utilization by soil microorganisms: a review. Soil Biol Biochem 42:2058–2067. https://doi.org/10.1016/j.soilbio.2010.08.021
Geisseler D, Scow KM (2014) Long-term effects of mineral fertilizers on soil microorganisms—a review. Soil Biol Biochem 75:54–63. https://doi.org/10.1016/j.soilbio.2014.03.023
Hartman WH, Richardson CJ (2013) Differential nutrient limitation of soil microbial biomass and metabolic quotients (qCO2): is there a biological stoichiometry of soil microbes? PLoS One 8:e57127. https://doi.org/10.1371/journal.pone.0057127
Hu C, Li SL, Qiao Y, Liu D, Chen Y (2015) Effects of 30 years repeated fertilizer applications on soil properties, microbes and crop yields in rice-wheat cropping systems. Exp Agric 51:355–369. https://doi.org/10.1017/S0014479714000350
Hu C, Xia X, Chen Y, Qiao Y, Liu D, Fan J, Li S (2019) Yield, nitrogen use efficiency and balance response to thirty-five years of fertilization in paddy rice-upland wheat cropping system. Plant Soil Environ 2019:55–62
Kuzyakov Y, Xu X (2013) Competition between roots and microorganisms for nitrogen: mechanisms and ecological relevance. New Phytol 198:656–669. https://doi.org/10.1111/nph.12235
Li P, Yang Y, Han W, Fang J (2014) Global patterns of soil microbial nitrogen and phosphorus stoichiometry in forest ecosystems. Glob Ecol Biogeogr 23:979–987. https://doi.org/10.1111/geb.12190
Lin S, Wang S, Si Y, Yang W, Zhu S, Ni W (2017) Variations in eco-enzymatic stoichiometric and microbial characteristics in paddy soil as affected by long-term integrated organic-inorganic fertilization. PLoS One 12:e0189908. https://doi.org/10.1371/journal.pone.0189908
Liu Y, Ge T, Zhu Z, Liu S, Luo Y, Li Y, Wang P, Gavrichkova O, Xu X, Wang J, Wu J, Guggenberger G, Kuzyakov Y (2019) Carbon input and allocation by rice into paddy soils: a review. Soil Biol Biochem 133:97–107. https://doi.org/10.1016/j.soilbio.2019.02.019
Luo Y, Dungait JJ, Zhao X, Brookes PC, Durenkamp M, Li G, Lin Q (2018) Pyrolysis temperature during biochar production alters its subsequent utilization by microorganisms in an acid arable soil. Land Degrad Dev. https://doi.org/10.1002/ldr.2846
Luo Y, Zhu Z, Liu S, Peng P, Xu J, Brookes P, Ge T, Wu J (2019) Nitrogen fertilization increases rice rhizodeposition and its stabilization in soil aggregates and the humus fraction. Plant Soil 445:125–135. https://doi.org/10.1007/s11104-018-3833-0
Magill AH, Aber JD (2000) Variation in soil net mineralization rates with dissolved organic carbon additions. Soil Biol Biochem 32:597–601. https://doi.org/10.1016/S0038-0717(99)00186-8
Mohanty S, Nayak AK, Kumar A, Tripathi R, Shahid M, Bhattacharyya P, Raja R, Panda BB (2013) Carbon and nitrogen mineralization kinetics in soil of rice-rice system under long term application of chemical fertilizers and farmyard manure. Eur J Soil Biol 58:113–121. https://doi.org/10.1016/j.ejsobi.2013.07.004
Mooshammer M, Wanek W, Zechmeister BS, Richter A (2014) Stoichiometric imbalances between terrestrial decomposer communities and their resources: mechanisms and implications of microbial adaptations to their resources. Front Microbiol 5:1–10. https://doi.org/10.3389/fmicb.2014.00022
Murphy J, Riley JP (1962) A modified of single solution method for the determination of phosphate in nature water. Anal Chim Acta 27:31–36. https://doi.org/10.1016/S0003-2670(00)88444-5
Murugan R, Kumar S (2013) Influence of long-term fertilisation and crop rotation on changes in fungal and bacterial residues in a tropical rice-field soil. Biol Fertil Soils 49:847–856. https://doi.org/10.1007/s00374-013-0779-5
Olsen S, Code C, Watanabe F, Dean L (1954) Estimation of available phosphorus in soils by extraction with sodium bicarbonate. Department of Agriculture, Washington, pp 1–9
Qiu H, Ge T, Liu J, Chen X, Hu Y, Wu J, Su Y, Kuzyakov Y (2018) Effects of biotic and abiotic factors on soil organic matter mineralization: experiments and structural modeling analysis. Eur J Soil Biol 84:27–34. https://doi.org/10.1016/J.EJSOBI.2017.12.003
Ren F, Sun N, Xu M, Zhang X, Wu L, Xu M (2019) Changes in soil microbial biomass with manure application in cropping systems: a meta-analysis. Soil Tillage Res 194:104291. https://doi.org/10.1016/j.still.2019.06.008
Schloter M, Nannipieri P, Sørensen SJ, van Elsas JD (2018) Microbial indicators for soil quality. Biol Fertil Soils 54:1–10. https://doi.org/10.1007/s00374-017-1248-3
Shi MS, Chang-yin LU, Hai-hou W, Xinwei Z, Meijuan J, Dong W (2015) Soil phosphorus dynamic, balance and critical P values in long-term fertilization experiment in Taihu Lake region, China. J Integr Agric 14:2446–2455. https://doi.org/10.1016/S2095-3119(15)61183-2
Singh JS, Gupta VK (2018) Soil microbial biomass: a key soil driver in management of ecosystem functioning. Sci Total Environ 634:497–500. https://doi.org/10.1016/j.scitotenv.2018.03.373
Soares M, Rousk J (2019) Microbial growth and carbon use efficiency in soil: links to fungal-bacterial dominance, SOC-quality and stoichiometry. Soil Biol Biochem 131:195–205. https://doi.org/10.1016/j.soilbio.2019.01.010
Stanford G, Smith SJ (1972) Nitrogen mineralization potentials of soils. Soil Sci Soc Am J Proc 36:465–472
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass C. Soil Biol Biochem 19:703–707. https://doi.org/10.1016/0038-0717(87)90052-6
Voroney RP, Brookes PC, Beyaert RP (2008) Soil microbial biomass C, N, P, and S. In: Carter MR, Gregorich EG (eds) Methods of analysis, 2nd edn. Taylor & Francis Group, LLC, Boca Raton, pp 637–665
Wang D, Zhu Z, Shahbaz M, Chen L, Liu S, Inubushi K, Wu J, Ge T (2019) Split N and P addition decreases straw mineralization and the priming effect of a paddy soil: a 100-day incubation experiment. https://doi.org/10.1007/s00374-019-01383-6
Wei X, Razavi BS, Hu Y, Xu X, Zhu Z, Liu Y, Kuzyakov Y, Li Y, Wu J, Ge T (2019) C/P stoichiometry of dying rice root defines the spatial distribution and dynamics of enzyme activities in root-detritusphere. Biol Fertil Soils 55:251–263. https://doi.org/10.1007/s00374-019-01345-y
Xu X, Pei J, Xu Y, Wang J (2020) Soil organic carbon depletion in global Mollisols regions and restoration by management practices: a review. J Soils Sediments 14:12–17. https://doi.org/10.1007/s11368-019-02557-3
Xu Y, Ding F, Gao X, Wang Y, Li M, Wang J (2019) Mineralization of plant residues and native soil carbon as affected by soil fertility and residue type. J Soils Sediments 19:1407–1415. https://doi.org/10.1007/s11368-018-2152-7
Yu Z, Chen L, Pan S, Li Y, Kuzyakov Y, Xu J, Brookes PC, Luo Y (2018) Feedstock determines biochar-induced soil priming effects by stimulating the activity of specific microorganisms. Eur J Soil Sci 69:521–534. https://doi.org/10.1111/ejss.12542
Zang H, Blagodatskaya E, Wang J, Xu X, Kuzyakov Y (2017) Nitrogen fertilization increases rhizodeposit incorporation into microbial biomass and reduces soil organic matter losses. Biol Fertil Soils 53:419–429. https://doi.org/10.1007/s00374-017-1194-0
Zhang W, Xu M, Wang X, Huang Q, Nie J, Li Z, Li S, Hwang SW, Lee KB (2012) Effects of organic amendments on soil carbon sequestration in paddy fields of subtropical China. J Soils Sediments 12:457–470. https://doi.org/10.1007/s11368-011-0467-8
Zhao FZ, Ren CJ, Han XH, Yang GH, Wang J, Doughty R (2018) Changes of soil microbial and enzyme activities are linked to soil C, N and P stoichiometry in afforested ecosystems. For Ecol Manag 427:289–295. https://doi.org/10.1016/j.foreco.2018.06.011
Zhou Z, Wang C, Jin Y (2017) Stoichiometric responses of soil microflora to nutrient additions for two temperate forest soils. Biol Fertil Soils 53:397–406. https://doi.org/10.1007/s00374-017-1188-y
Zhu Z, Ge T, Luo Y, Liu S, Xu X, Tong C, Shibistova O, Guggenberger G, Wu J (2018) Microbial stoichiometric flexibility regulates rice straw mineralization and its priming effect in paddy soil. Soil Biol Biochem 121:67–76. https://doi.org/10.1016/j.soilbio.2018.03.003
Acknowledgments
We gratefully acknowledge the financial support provided by the National Natural Science Foundation of China (Grant numbers 41877105, 41907093) and The Fundamental Research Funds for Central Non-profit Scientific Institution (1610132019044, 1610132019013). We also acknowledged the anonymous reviewers for their constructive suggestions on this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
No informed or ethical consent was required for this research.
Additional information
Responsible editor: Weixin Ding
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 22 kb)
Rights and permissions
About this article
Cite this article
Ashraf, M.N., Hu, C., Wu, L. et al. Soil and microbial biomass stoichiometry regulate soil organic carbon and nitrogen mineralization in rice-wheat rotation subjected to long-term fertilization. J Soils Sediments 20, 3103–3113 (2020). https://doi.org/10.1007/s11368-020-02642-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-020-02642-y