Abstract
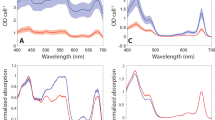
Photosynthetic organisms alter their photosynthetic pigment composition in response to changes in growth irradiance1–3. Photo-adaptation maximizes light-harvesting when photon flux densities are low, and minimizes photo-oxidative damage to the photosynthetic machinery at high light levels. In chlorophytes, the major light-harvesting antenna is light-harvesting complex II (LHC II), a family of proteins binding chlorophyll a (Chl a), Chl b, and carotenoids, and accounting for 40–60% of total cell chlorophyll4. LHC II is associated principally with photosystem II, with reversible phosphorylation of LHC II regulating short-term adjustments in energy distribution to photosystem I5. Previous studies on green algae and higher plants have emphasized the longer-term adaptive importance of the inverse relationship between growth irradiance and the proportion of total cellular chlorophyll associated with LHC II6–8. In higher plants the pigment composition of LHC II appears to be highly conserved, with Chl a/Chl b ratios between 1.0 and 1.2 (ref 7). In green algae, the pigment ratio of LHC II is more variable and values between 0.7 and 2.7 have been reported9–11. We report here that in the unicellular marine chlorophyte, Dunaliella tertiolecta, the ratio is actually variable. Photoadapta-tion to high irradiance involves changes in the average composition and behaviour of LHC II; specifically, the Chl b content per polypeptide is halved and the efficiency of excitation transfer from carotenoid to Chl a declines. The result is a novel mechanism for regulating the effective absorption cross-section of photosystem II.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
1. Drews, G. Trends Biochem. Sci. 11, 255–257 (1981).
2. Falkowski, P. G. in Primary Productivity in the Sea (ed. Falkowski, P. G.) 99–119 (Plenum, New York, 1980).
3. Boardman, N. K. A. Rev. Pi Physiol. 28, 355–377 (1977).
4. Bennett, J. Biochem. J. 212, 1–13 (1983).
5. Staehelin, L. A. & Arntzen, C. J. J. cell Biol. 97, 1327–1337 (1983).
6. Kan, K.–S. & Thornber, J. P. PL Physiol. 57, 47–52 (1976).
7. Anderson, J. M. A Rev. PI. Physiol. 37, 93–136 (1986).
8. Leong, T.–Y. & Anderson, J. M. Biochem. biophys. Acta 850, 57–63 (1986).
9. Apel, K. Brookhaven Symp. Biol. 28, 149–161 (1977).
10. Cunningham, F. & Shiff, J. A. PL Physiol. 80, 223–230 (1986).
11. Anderson, J. M., Waldron, J. C. & Thorne, S. W. PL Sci. Lett. 17, 149–151 (1980).
12. Leong, T.–Y. & Anderson, J. M. Photosynth. Res. 5, 105–115 (1984).
13. Herron, H. A. & Mauzerall, D. PL Physiol. 50, 141–148 (1972).
14. Bradbury, M. & Baker, N. R. Biochim. biophys. Acta 635, 562–551 (1981).
15. Sukenik, A., Bennett, J. & Falkowski, P. G. Biochim. biophys. Acta (submitted).
16. van Metter, R. L. Biochim. biophys. Acta 462, 642–658 (1977).
17. Falkowski, P. G., Fujita, Y., Ley, A. & Mauzerall, D. PL Physiol. 81, 310–312 (1986).
18. Lee, P. & Horton, P. FEBS Lett. 162, 81–84 (1983).
19. Kyle, D. J. Photochem. Photobiol. 41, 107–116 (1985).
20. Kyle, D. J., Ohad, I. & Arntzen, C. J. Proc. natn. Acad. Sci. U.S.A 81, 4070–4074 (1984).
21. Schuster, G., Dewit, M., Staehelin, L. A. & Ohad, I. J. cell Biol. 103, 71–80 (1986).
22. Michel, H. P., Tellenbach, M. & Boschetti, A. Biochim. biophys. Acta 725, 417–424 (1983).
23. Darr, S. C., Somerville, S. C. & Arntzen, C. J. J. Cell Biol. 103, 733–740 (1986).
24. Falkowski, P. G. Photosynthetica 18, 62–62 (1984).
25. Bennett, J., Jenkins, G. I. & Hartley, M. R. J. cell. Biochem. 25, 1–13 (1984).
26. Williams, R. S. & Ellis, R. J. FEBS Lett. 203, 295–300 (1986).
27. Williams, R. S., Shaw, E. K., Sieburth, L. E. & Bennett, J. Meth. Enzym. 118,338–352 (1986).
28. Falkowski, P. G., Wyman, K. D., Ley, A. C. & Mauzerall, D. C. Biochim. biophys. Acta 849, 183–192 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sukenik, A., Wyman, K., Bennett, J. et al. A novel mechanism for regulating the excitation of photosystem II in a green alga. Nature 327, 704–707 (1987). https://doi.org/10.1038/327704a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/327704a0
This article is cited by
-
Chlorella vulgaris integrates photoperiod and chloroplast redox signals in response to growth at high light
Planta (2019)
-
Global transcriptome analyses provide evidence that chloroplast redox state contributes to intracellular as well as long-distance signalling in response to stress and acclimation in Arabidopsis
Photosynthesis Research (2016)
-
Corals as light collectors: an integrating sphere approach
Coral Reefs (2005)
-
A light-harvesting siphonax-anthin-chlorophylla/b-protein complex of marine green alga,Bryopsis corticulans
Chinese Science Bulletin (2004)
-
Chloroplast avoidance movement reduces photodamage in plants
Nature (2002)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.