Abstract
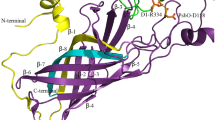
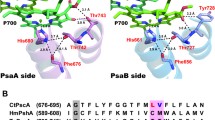
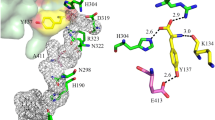
The psaA and psaB genes of the chloroplast genome in oxygenic photosynthetic organisms code for the major peptides of the Photosystem 1 reaction center. A heterodimer of the two polypeptides PsaA and PsaB is thought to bind the reaction center chlorophyll, P700, and the early electron acceptors A0, A1 and Fe-SX. Fe-SX is a 4Fe4S center requiring 4 cysteine residues as ligands from the protein. As PsaA and PsaB have only three and two conserved cysteine residues respectively, it has been proposed by several groups that Fe-SX is an unusual inter-peptide center liganded by two cysteines from each peptide. This hypothesis has been tested by site directed mutagenesis of PsaA residue C575 and the adjacent D576. The C575D mutant does not assemble Photosystem 1. The C575H mutant contains a photoxidisable chlorophyll with EPR properties of P700, but no other Photosystem 1 function has been detected. The D576L mutant assembles a modified Photosystem 1 in which the EPR properties of the Fe-SA/B centers are altered. The results confirm the importance of the conserved cysteine motif region in Photosystem 1 structure.
Similar content being viewed by others
References
Armstrong FA, George SJ, Cammack R, Hatchikian EC and Thomson AJ (1989) Electrochemical and spectroscopic characterisation of the 7 Fe form of ferredoxin III from Desulphovibrio africanus. Biochem J 264: 265–274
Britt RD, Sauer K, Klein MP, Knaff DB, Kriauciunas A, Yu CA, Yu L and Malkin R (1991) ESEEM spectroscopy supports the suggested coordination of two histidine ligands to the Reiske Fe-S center. Biochemistry 30: 1982–1901
Delepelaire P and Chua N-H (1979) Lithium dodecyl sulfate/polyacrylamide gel electrophoresis of thylakoid membranes at 4 °C: Characterisation of two additional chlorophyll a-protein complexes. Proc Natl Acad Sci (USA) 76: 111–115
Diner BA and Wollman FA (1980) Isolation of highly active Photosystem II complexes of C. reinhardtii. Eur J Biochem 110: 521–5261
Evans MCW and Nugent JHA (1993) Structure and function of the reaction center cofactors of oxygenic organisms. In: Norris JR and Diesenhoffer J (eds) The Photosynthetic Reaction Center, Vol 1, pp 391–415. Academic Press, New York
Evans MCW, Hallahan BJ, Hanley JA, Heathcote P, Gumpel NJ and Purton S (1995) Isolation and characterization of photosynthetic reaction centers from eukaryotic organisms. In: Cooper C and Brown G (eds) Bioenergetics: A Practical Approach, pp 183–213. IRL Press, Oxford, UK
Fish LE, Kück U and Bogorad L (1985) Two partially homologous adjacent light inducible chloroplast genes encoding polypeptides of the P700 chlorophyll a protein complex of Photosystem I. J Biol Chem 260: 1413–1421
Golbeck JH (1992) Structure and Function of Photosystem-I. Ann Rev Plant Physiol Plant Mol Biol 43: 293–324
Golbeck JH and Cornelius JM (1986) Photosystem 1 charge separation in the absence of Centers A and B. 1. Optical characterisation of Center ‘A2’ and evidence for its association with a 64 kDa peptide. Biochim Biophys Acta 549: 16–24
Goldschmidt-Clermont M (1991) Transgenic expression of aminoglycoside adenine transferase in the chloroplast: A selectable marker for site-directed transformation of Chlamydomonas. Nucl Acids Res 19: 4083–4089
Hanley JA, Kear J, Bredenkamp G, Li G, Heathcote P and Evans MCW (1992) Biochemical evidence for the role of the bound iron-sulphur centers A and B in NADP reduction by Photosystem 1. Biochim Biophys Acta 1099: 152–156
Harris EH (1989) The Chlamydomonas Sourcebook. A Comprehensive Guide to Biology and Laboratory Use. Academic Press, San Diego
Höj PB and Møller BL (1986) The 110 kDa reaction center protein of Photosystem 1, chlorophyll a protein 1, is an iron-sulphur protein. J Biol Chem 261: 14292–14300
Höj PB, Svendsen IB, Scheller HV and Moller BL (1987) Identification of a chloroplast-encoded 9-kDa polypeptide as a 2[4Fe-4S] protein carrying centers A and B of Photosystem I. J Biol Chem 262: 12676–12684
Krauss N, Hinrichs W, Witt I, Fromme P, Pritzkow W, Dauter Z, Betzel C, Wilson KS, Witt HT and Saenger W (1993) 3-Dimensional Structure of System-I of Photosynthesis at 6 Angstrom Resolution. Nature 361: 326–331
Kück U, Choquet Y, Schneider M, Dron M and Bennoun P (1987) Structural and transcriptional analysis of two homologous genes for the P700 chlorophyll a-apoproteins in Chlamydomonas reinhardtii: Evidence for in vivo trans-splicing. EMBO J 6: 2185–2195
Li N, Zhao J, Warren PV, Warden JT, Bryant DA and Golbeck JH (1991) PsaD is required for stable binding of PsaC to the Photosystem 1 core protein of Synechococcus sp. PCC 6301. Biochemistry 30: 7863–7872
Moenne-Loccoz P, Heathcote P, MacLachlan DJ, Berry MC, Davis IH Evans MCW (1994) The path of electron transfer in Photosystem 1: Direct evidence of forward electron transfer from A1 to Fe-SX. Biochemistry 33: 10037–10042
Nugent JHA and Evans MCW (1979) Light induced signals at cryogenic temperatures in subchloroplast particles enriched in Photosystem II. FEBS Letts 101: 101–104
O'Connor HE, Stevens DR, Ruffle SV, Nugent JHA and Purton S (1995) An improved procedure for the isolation of chloroplast DNA from Chlamydomonas reinhardtii. Plant Mol Biol Rep 11: 207–211
Rochaix J-D, Mayfield S, Goldschmidt-Clermont M and Erickson J (1988) Molecular biology of Chlamydomonas reinhardtii. In: Shaw CH (ed) Plant Molecular Biology: A Practical Approach, pp 253–275. IRL Press, Oxford, UK
Rodday SM, Jun S-S and Biggins J (1993) Interaction of the FA, FB containing subunit with the Photosystem 1 core heterodimer. Photosynth Res 36: 1–9
Rodday SM, Schulz R, Mcintosh L and Biggins J (1994) Structure-function studies on the interaction of PsaC with the Photosystem I heterodimer. Photosynth Res 42: 185–190
Rodday SM, Webber AN, Bingham SE and Biggins J (1995) Evidence that the FX domain in Photosystem 1 interacts with the subunit PsaC: Site directed changes in PsaB destabilise the subunit interaction in Chlamydomonas reinhardtii
Sanford JC (1988) The biolistic process. Trends Biotechnol 6: 299–302
Sanford JC, Smith FD and Russell JA (1993) Optimizing the biolistic process for different biological applications. Methods Enzymol 217: 483–509
Smart LB and McIntosh L (1991) Genetic inactivation of the psaB gene in Synechocystis sp. PCC 6803 disrupts assembly of Photosystem I. Plant Mol Biol 21: 177–180
Smart LB, Warren PV, Golbeck JH and McIntosh L (1993) Mutational Analysis of the Structure and Biogenesis of the Photosystem-I Reaction Center in the Cyanobacterium Synechocystis Sp-PCC-6803 Proc Natl Acad Sci USA 90: 1132–1136
Takahashi Y, Goldschmidt-Clermont M, Soen S-Y, Franzen LG and Rochaix J-D (1991) Directed chloroplast transformation in Chlamydomonas reinhardtii: Insertional inactivation of the psaC gene encoding the iron sulfur protein destabilizes Photosystem I. EMBO J 10: 2033–2040
Warren PV, Smart LB, McIntosh L and Golbeck JH (1993) Site-directed conversion of systeine-565 to serine in PsaB of Photosystem 1 results in the assembly of [3Fe-4S] and [4Fe-4S] clusters in FX. A mixed ligand [4Fe-4S] cluster is capable of electron transfer to FA and FB. Biochemistry 32: 4411–4419
Webber AN, Gibbs PB, Ward JB and Bingham SE (1993) Site-directed mutagenesis of the Photosystem I reaction center in chloroplasts. The proline-cysteine motif. J Biol Chem 268: 12990–12995
Wynn RM and Malkin R (1988) Characterisation of an isolated chloroplast membrane Fe-S protein and its identification as the PS 1 Fe-SA/Fe-SB binding protein. FEBS Letts 229: 293–2971
Author information
Authors and Affiliations
Additional information
Dedicated to the memory of Daniel I. Arnon.
Rights and permissions
About this article
Cite this article
Hallahan, B.J., Purton, S., Ivison, A. et al. Analysis of the proposed Fe-SX binding region of Photosystem 1 by site directed mutation of PsaA in Chlamydomonas reinhardtii . Photosynth Res 46, 257–264 (1995). https://doi.org/10.1007/BF00020438
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00020438