Abstract
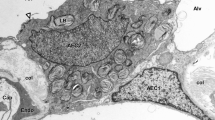
The cellular repair process of injured tracheal epithelium is described for sheep after exposure to toxic smoke containing high concentrations of acrolein. Fourteen fasted 3–4-year-old ewes had a portion of their cervical trachea exposed to cotton smoke for 20 min and then were sacrificed at various time intervals ranging from 1 to 22 days after exposure. Within 1 day of injury, columnar epithelium sloughed intact from the trachea with a concomitant reduction of nearly 35% in the basal cell population. At 2 days of recovery, the cellularity of the epithelium had increased and mitotic figures were observed in some tracheal epithelial and gland cells. By 8 days, undifferentiated hyperplastic cells increased to 30/100 µm, differentiated nonciliated columnar cells first appeared, and the basal cell population returned to a normal count of 13 cells/100µm. Thirteen days after exposure, the undifferentiated hyperplastic cell population had declined to 7 cells/100/ µm, nonciliated columnar cells were at control values, and some ciliated cells were identified. At 18 and 22 days, epithelium was normal in appearance and the count was 13 cells/100µm. Data suggest that because the columnar epithelium sloughs intact with the cilia remaining active, toxic smoke may affect their attachment to the basal lamina. Furthermore, the regeneration process involves differentiation of hyperplastic cells in which they elongate down to the basal lamina, thus re-establishing the integrity of tall, epithelium in the sheep trachea.
Similar content being viewed by others
References
Abdi S, Evans MJ, Cox RA, Lubbesmeyer H, Herndon DN, Traber DL (1990) Inhalation injury to tracheal epithelium in an ovine model of cotton smoke exposure—early phase (30 minutes). Am Rev Respir Dis 142:1436–1439
Astry CL, Jakab CJ (1983) Effects of acrolein exposure on pulmonary antibacterial defenses. Toxicol Appl Pharmacol 67:49–54
Barrow RE, Morris SE, Basadre JO, Herndon DN (1990) Selective permeability changes in the lungs and airways of sheep after toxic smoke inhalation. J Appl Physiol 68:2165–2170
Barrow RE, Morris SE, Linares HA, Herndon DN (1991) Tracheal venous blood and lymph collection: a model to study airway injury in sheep. J Appl Physiol 70(4):1645–1649
Beauchamp RQ, Jr., Morgan KT, Kligerman AD, Andelkovich DA, Heck HA (1985) A critical review of the literature on acrolein toxicity. CRC Crit Rev Toxicol 14:309–380
Boucher RC, Van Scott MR, Willumsen N, Stutts MJ (1988) Mechanisms and cell biology of airway epithelial injury. Am Rev Respir Dis 138:S41-S44
Calhoun KH, Deskin RW, McCrachen MM, Hokanson JA, Garza C, Nichols RJ Jr, Herndon DN (1988) Long-term airway sequelae in a pediatric burn population. Laryngoscope 98:721–725
Carson JL, Collier AM, Clyde WA (1979) Ciliary membrane alterations occurring in experimental Mycoplasma pneumoniae infection. Science 206:349–351
Cosa DL, Kutzman RL, Lehmann JR, Drew RT (1986) Altered lung function and structure in the rat after subchronic exposure to acrolein. Am Rev Respir Dis 133:286–291
Ecnerbom B, Lindholm CE, Alexopoulos C (1986) Airway lesions caused by prolonged intubation with standard and with anatomically shaped tracheal tubes. A post-mortem study. Acta Anaesth Scand 30:366–373
Evans MJ (1982) Cell death and cell renewal in small airways and alveoli. In: Mechanisms in respiratory toxicology, vol. 1. Boca Raton, CRC Press, pp 189–218
Evans MJ, Shami SG (1989) Lung cell kinetics. In: Lung cell biology. Marcel Dekker, New York, pp 1–36
Evans MJ, Cox RA, Shami SG, Wilson B, Plopper CG (1989) The role of basal cells in attachment of columnar cells to the basal lamina of trachea. Am J Respir Cell Mol Biol 1:463–469
Evans MJ, Cox RA, Shami SG, Plopper CG (1990) Junctional adhesion mechanisms in airway basal cells. Am J Respir Cell Mol Biol 3:341–347
Kauffman SL (1980) Cell proliferation in the mammalian lung. Int Rev Exp Pathol 22:131–191
Keenan KP, Combs JW, McDowell EM (1982) Regeneration of hamster tracheal epithelium after mechanical injury. 1. Focal lesions: quantitative morphologic study of cell proliferation. Virchows Arch (B) 41:193–214
Knowles MR, Gatzy JT, Boucher RC (1981) Increased bioelectric potential differences across respiratory epithelia in cystic fibrosis. N Eng J Med 305:1489–1495
Linares HA, Herndon DN, Traber DL (1989) Sequence of morphologic events in experimental smoke inhalation. J Burn Care Rehab 10:27–37
Lund T, Goodwin CW, McManus WF, Shirani KZ, Stallings RJ, Mason AD, Pruitt BA Jr (1985) Upper airway sequelae in burn patients requiring endotracheal intubation or tracheostomy. Ann Surg 201(3):374–381
Man SFP, Hubert CW (1988) Airway repair and adaptation to inhalation injury. In: Pathophysiology and treatment of inhalation injury. New York, Marcel Dekker, pp 1–47
Shami SG, Thibodeaux LA, Kennedy AR, Little JB (182) Recovery from ozone-induced injury in the lungs of Syrian golden hamster. Exp Mol Pathol 36:57–71
Author information
Authors and Affiliations
Additional information
Offprint requests to: R. E. Barrow
Rights and permissions
About this article
Cite this article
Barrow, R.E., Wang, CZ., Cox, R.A. et al. Cellular sequence of tracheal repair in sheep after smoke inhalation injury. Lung 170, 331–338 (1992). https://doi.org/10.1007/BF00177579
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00177579