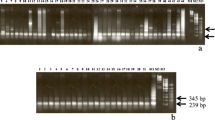
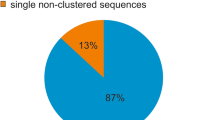
Abstract
Deschampsia antactica E. Desv. is one of the only two native vascular plants of Antarctica, having a disjunct distribution with South America. Its presence in different environmental conditions turns it into an interesting evolution model, particularly for genomic evolutionary studies. The repetitive DNA is a genome component that cause important changes in genome size and chromosome organization, and therefore, its variation is very important in group’s delimitation. Some tandem repetitive DNA sequences, known as satellite DNA (satDNAs) are shared between many groups of Poaceae (e.g., of these are the CON1, CON2, COM1, and COM2 sequences) highlighting its evolutionary component. This study aims to identify, classify, and characterize repetitive elements in the D. antarctica genome by clustering analysis of genome sequences, focusing on the CON1, CON2, COM1, and COM2. Repetitive DNA represented about 73.3% of the D. antarctica genome. All studied populations presented loci for the studied satDNAs but the distribution pattern showed differences that seem to be related to the geographic distribution. The analysis of CON/COM sequences in D. antarctica contributes to the understanding of these elements in Poaceae genomes and highlights the importance of changes in chromosome organization of repetitive DNA in populations with fragmented geographical distribution. The distribution of such chromosome changes may both reflect the process of colonization of D. antarctica in Antarctica and explain some evolutionary processes of differentiation in Deschampsia species complex in the Patagonia, which is still unresolved with other DNA sequences.






Similar content being viewed by others
References
Alberdi M, Bravo LA, Gutiérrez A et al (2002) Ecophysiology of Antarctic vascular plants. Physiol Plant 115:479–486
Alix K, Baurens F-C, Paulet F et al (1998) Isolation and characterization of a satellite DNA family in the Saccharum complex. Genome 41:854–864
Amosova AV, Bolsheva NL, Samatadze TE et al (2015) Molecular cytogenetic analysis of Deschampsia antarctica Desv. (Poaceae), Maritime Antarctic. PLoS ONE 10:e0138878
Aversano R, Contaldi F, Ercolano MR et al (2015) The Solanum commersonii Genome sequence provides insights into adaptation to stress conditions and genome evolution of wild potato relatives. Plant Cell 27:954–968
Bennett MD, Leitch IJ (2012) Plant DNA C-values database. http://data.kew.org/cvalues. Accessed 24 April 2017
Bennett MD, Smith JB, Smith R (1982) DNA amounts of angiosperms from the Antarctic and South Georgia. Environ Exp Bot 22:307–318
Benson G (1999) Tandem repeats finder: a program to analyze DNA sequences. Nucleic Acids Res 27:573–580
Bostock C (1980) A function for satellite DNA? Trends Biochem Sci 5:117–119
Brandes A, Röder MS, Ganal MW (1995) Barley telomeres are associated with two different types of satellite DNA sequences. Chromosome Res 3:315–320
Bravo LA, Ulloa N, Zuñiga GE et al (2001) Cold resistance in Antarctic angiosperms. Physiol Plant 111:55–65
Cardone S, Sawatani P, Rush P et al (2008) Karyological studies in Deschampsia antarctica Desv. (Poaceae). Polar Biol 32:427–433
Chiapella J (2007) A molecular phylogenetic study of Deschampsia (Poaceae: Aveneae) inferred from nuclear ITS and plastid trnL sequence data: support for the recognition of Avenella and Vahlodea. Taxon 56:55–64
Chiapella J, Zuloaga FO (2010) A Revision of Deschampsia, Avenella, and Vahlodea (Poaceae, Poeae, Airinae) in South America. Ann Mo Bot Gard 97:141–162
Chwedorzewska KJ (2006) Preliminary genetic study on species from genus Deschampsia from Antarctic (King George I.) and Arctic (Spitsbergen). Polar Biosci 19:142–147
Crooks GE, Hon G, Chandonia J-M, Brenner SE (2004) WebLogo: a sequence logo generator. Genome Res 14:1188–1190
Derks MFL, Smit S, Salis L et al (2015) The genome of winter moth (Operophtera brumata) provides a genomic perspective on sexual dimorphism and phenology. Genome Biol Evol 7:2321–2332
Dolezel J, Bartos J, Voglmayr H, Greilhuber J (2003) Nuclear DNA content and genome size of trout and human. Cytometry A 51:127–129
Dover GA (1986) Molecular drive in multigene families: How biological novelties arise, spread and are assimilated. Trends Genet 2:159–165
Emadzade K, Jang T-S, Macas J et al (2014) Differential amplification of satellite PaB6 in chromosomally hypervariable Prospero autumnale complex (Hyacinthaceae). Ann Bot 114:1597–1608
Evtushenko EV, Vershinin AV (2010) Heterogeneous organization of a tandem repeat family in subtelomeric heterochromatin of rye. Russ J Genet 46:1074–1076
Fasanella M, Premoli AC, Urdampilleta JD, González ML, Chiapella J (2017) How did a grass reach Antarctica? The Patagonian connection of Deschampsia antarctica (Poaceae). Bot J Linn Soc (in press)
Feitoza L, Guerra M (2011) Different types of plant chromatin associated with modified histones H3 and H4 and methylated DNA. Genetica 139:305–314
Flavell RB (1986) Repetitive DNA and chromosome evolution in plants. Philos Trans R Soc Lond B 312:227–242
Fry K, Salser W (1977) Nucleotide sequences of HS-alpha satellite DNA from kangaroo rat Dipodomys ordii and characterization of similar sequences in other rodents. Cell 12:1069–1084
Garrido-Ramos MA (2015) Satellite DNA in Plants: more than Just Rubbish. Cytogenet Genome Res 146:153–170
Gidekel M, Destefano-Beltrán L, García P et al (2003) Identification and characterization of three novel cold acclimation-responsive genes from the extremophile hair grass Deschampsia antarctica Desv. Extremophiles 7:459–469
González ML, Urdampilleta JD, Fasanella M et al (2016) Distribution of rDNA and polyploidy in Deschampsia antarctica E. Desv. in Antarctic and Patagonic populations. Polar Biol 39:1663–1677
Grebenstein B, Grebenstein O, Sauer W, Hemleben V (1995) Characterization of a highly repeated DNA component of perennial oats (Helictotrichon, Poaceae) with sequence similarity to a A-genome-specific satellite DNA of rice (Oryza). Theor Appl Genet 90:1101–1105
Grebenstein B, Grebenstein O, Sauer W, Hemleben V (1996) Distribution and complex organization of satellite DNA sequences in Aveneae species. Genome 39:1045–1050
Guerra M (1988) Introdução à citogenética geral. Guanabara Koogan
Hemleben V, Kovarik A, Torres-Ruiz RA et al (2007) Plant highly repeated satellite DNA: molecular evolution, distribution and use for identification of hybrids. Syst Biodivers 5:277–289
Holderegger R, Stehlik I, Lewis Smith RI, Abbott RJ (2003) Populations of Antarctic hairgrass (Deschampsia antarctica) show low genetic diversity. Arct Antarct Alp Res 35:214–217
Ištvánek J, Jaroš M, Křenek A, Řepková J (2014) Genome assembly and annotation for red clover (Trifolium pratense; Fabaceae). Am J Bot 101:327–337
Jiang J, Hulbert SH, Gill BS, Ward DC (1996) Interphase fluorescence in situ hybridization mapping: a physical mapping strategy for plant species with large complex genomes. Mol Gen Genet 252:497–502
John UP, Polotnianka RM, Sivakumaran KA et al (2009) Ice recrystallization inhibition proteins (IRIPs) and freeze tolerance in the cryophilic Antarctic hair grass Deschampsia antarctica E. Desv. Plant Cell Environ 32:336–348
King M (1995) Species evolution: the role of chromosome change. Cambridge University Press, Cambridge
Komarkova V, Poncet S, Poncet J (1985) Two native Antarctic vascular plants, Deschampsia antarctica and Colobanthus quitensis: a new southernmost locality and other localities in the Antarctic Peninsula area. Arctic Alpine Res 17:401
Komarkova V, Poncet S, Poncet J (1990) Additional and revisited localities of vascular plants Deschampsia antarctica Desv. and Colobanthus quitensis (Kunth) Bartl. in the Antarctic Peninsula area. Arctic Alpine Res 22:108
Křivánková A, Kopecký D, Stočes Š et al (2017) Repetitive DNA: a versatile tool for karyotyping in Festuca pratensis Huds. Cytogenet Genome Res 151:96–105
Kubis S, Schmidt T, Heslop-Harrison JS (1998) Repetitive DNA elements as a major component of plant genomes. Ann Bot 82:45–55
Lamb JC, Danilova T, Bauer MJ et al (2007) Single-gene detection and karyotyping using small-target fluorescence in situ hybridization on maize somatic chromosomes. Genetics 175:1047–1058
Lane Rayburn A, Gill BS (1986) Isolation of a D-genome specific repeated DNA sequence from Aegilops squarrosa. Plant Mol Biol Rep 4:102–109
Lee J, Kang Y, Shin SC et al (2014) Combined analysis of the chloroplast genome and transcriptome of the Antarctic vascular plant Deschampsia antarctica Desv. PLoS ONE 9:e92501
Levin DA (2002) The role of chromosomal change in plant evolution. Oxford University Press, Ney York
López-Flores I, Garrido-Ramos MA (2012) The repetitive DNA content of eukaryotic genomes. Genome Dyn 7:1–28
Macas J, Mészáros T, Nouzová M (2002) PlantSat: a specialized database for plant satellite repeats. Bioinformatics 18:28–35
Macas J, Neumann P, Navrátilová A (2007) Repetitive DNA in the pea (Pisum sativum L.) genome: comprehensive characterization using 454 sequencing and comparison to soybean and Medicago truncatula. BMC Genom 8:427
Margulies M, Egholm M, Altman WE et al (2005) Genome sequencing in microfabricated high-density picolitre reactors. Nature 437:376–380
Mehrotra S, Goyal V (2014) Repetitive sequences in plant nuclear DNA: types, distribution, evolution and function. Genom Proteom Bioinform 12:164–171
Metzlaff M, Troebner W, Baldauf F et al (1986) Wheat specific repetitive DNA sequences—construction and characterization of four different genomic clones. Theor Appl Genet 72:207–210
Montiel P, Smith A, Keiller D (1999) Photosynthetic responses of selected Antarctic plants to solar radiation in the southern maritime Antarctic. Polar Res 18:229–235
Moore DM (1970) Studies in Colobanthus Quitensis (Kunth) Bartl. and Deschampsia antarctica Desv. II. Taxonomy, distribution and relationships. Br Antarct Surv Bull 23:63–80
Mosyakin SL, Bezusko LG, Mosyakin AS (2007) Origins of native vascular plants of Antarctica: comments from a historical phytogeography viewpoint. Tsitol Genet 41:54–63
Murray BG, De Lange PJ, Ferguson AR (2005) Nuclear DNA variation, chromosome numbers and polyploidy in the endemic and indigenous grass flora of New Zealand. Ann Bot 96:1293–1305
Navrotska DO, Twardovska MO, Andreev IO et al (2014) New forms of chromosome polymorphism in Deschampsia antarctica Desv. from the Argentine islands of the Maritime Antarctic region. Ukrainian Antarct J 13:185–191
Novák P, Neumann P, Macas J (2010) Graph-based clustering and characterization of repetitive sequences in next-generation sequencing data. BMC Bioinform 11:378
Novák P, Neumann P, Pech J et al (2013) RepeatExplorer: a galaxy-based web server for genome-wide characterization of eukaryotic repetitive elements from next-generation sequence reads. Bioinformatics 29:792–793
Novák P, Ávila Robledillo L, Koblížková A et al (2017) TAREAN: a computational tool for identification and characterization of satellite DNA from unassembled short reads. Nucleic Acids Res. doi:10.1093/nar/gkx257
Parnikoza IY, Maidanuk DN, Kozeretska IA (2007a) Are Deschampsia antarctica Desv. and Colobanthus quitensis (Kunth) Bartl. migratory relicts? Tsitol Genet 41:36–40
Parnikoza IY, Miryuta NY, Maidanyuk DN et al (2007b) Habitat and leaf cytogenetic characteristics of Deschampsia antarctica Desv. in the Maritime Antarctica. Polar Sci 1:121–128
Plohl M (2005) The species-specificity and evolution of satellite DNAs with emphasis on satellite DNAs in tenebrionid beetles. Entomol Croat 9:85–96
Plohl M, Meštrović N, Mravinac B (2012) Satellite DNA evolution. Genome Dyn 7:126–152
Premoli AC, Mathiasen P, Acosta CM, Ramos VA (2012) Phylogeographically concordant chloroplast DNA divergence in sympatric Nothofagus ss How deep can it be? New Phytol 193:261–275
Reddy AS, Kiefer-Meyer MC, Delseny M (1993) Characterization of new variants of a satellite DNA from Oryza officinalis, specific for the CC genome of wild rice. Genome 36:750–761
Röser M, Winterfeld G, Döring E, Schneider J (2014) Chromosome evolution in grass tribes Aveneae/Poeae (Poaceae): insights from karyotype structure and molecular phylogeny. Schlechtendalia 28:1–21
Ruhland CT, Day TA (2000) Effects of ultraviolet-B radiation on leaf elongation, production and phenylpropanoid concentrations of Deschampsia antarctica and Colobanthus quitensis in Antarctica. Physiol Plant 109:244–251
Sanmiguel P, Bennetzen JL (1998) Evidence that a recent increase in maize genome size was caused by the massive amplification of intergene retrotransposons. Ann Bot 82:37–44
Schwarzacher T (2003) Meiosis, recombination and chromosomes: a review of gene isolation and fluorescent in situ hybridization data in plants. J Exp Bot 54:11–23
Schwarzacher T, Heslop-Harrison P et al (2000) Practical in situ hybridization. BIOS Scientific Publishers Ltd, Oxford
Sharma S, Raina SN (2005) Organization and evolution of highly repeated satellite DNA sequences in plant chromosomes. Cytogenet Genome Res 109:15–26
Stergianou KK (1989) Habit differentiation and chromosome evolution in Pleione (Orchidaceae). Plant Syst Evol 166:253–264
Sýkorová E, Fajkus J, Ito M, Fukui K (2001) Transition between two forms of heterochromatin at plant subtelomeres. Chromosome Res 9:309–323
Tiwari VK, Wang S, Danilova T et al (2015) Exploring the tertiary gene pool of bread wheat: sequence assembly and analysis of chromosome 5 mg of Aegilops geniculata. Plant J 84:733–746
Trifonov EN (1989) The multiple codes of nucleotide sequences. Bull Math Biol 51:417–432
Ugarković Đ (2008) Satellite DNA libraries and centromere evolution. The Open Evolution Journal 2:1–6
van de Wouw M, van Dijk P, Huiskes A (2008) Regional genetic diversity patterns in Antarctic hairgrass (Deschampsia antarctica Desv.). J Biogeogr 35:365–376
Vershinin AV, Schwarzacher T, Heslop-Harrison JS (1995) The large-scale genomic organization of repetitive DNA families at the telomeres of rye chromosomes. Plant Cell 7:1823–1833
Vieira RC, Mantovani A (1995) Anatomia foliar de Deschampsia antarctica Desv. (Gramineae). Rev Brasil Bot 18:207–220
Vittorazzi SE, Lourenço LB, Recco-Pimentel SM (2014) Long-time evolution and highly dynamic satellite DNA in leptodactylid and hylodid frogs. BMC Genet 15:111
Wang W, Ma L, Becher H et al (2016) Astonishing 35S rDNA diversity in the gymnosperm species Cycas revoluta Thunb. Chromosoma 125:683–699
Weising K, Nybom H, Pfenninger M et al (2005) DNA fingerprinting in plants: principles, methods, and applications, 2nd edn. CRC Press, Boca Raton
Winterfeld G, Röser M (2007) Chromosomal localization and evolution of satellite DNAs and heterochromatin in grasses (Poaceae), especially tribe Aveneae. Plant Syst Evol 264:75
Wolf PG, Sessa EB, Marchant DB et al (2015) An exploration into fern genome space. Genome Biol Evol 7:2533–2544
Acknowledgements
The authors are grateful to CONICET, ANPCyT-FONCyT, and SECyT-UNC for financial support and to Dirección Nacional del Antártico and the personnel of the Carlini Station for logistic support for fieldwork in Antarctica. This work was funded by project PICTO 2010–0095 (ANPCyT-DNA) and the Mrs. Jorgelina Brasca for the revision of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
González, M.L., Chiapella, J.O. & Urdampilleta, J.D. Characterization of some satellite DNA families in Deschampsia antarctica (Poaceae). Polar Biol 41, 457–468 (2018). https://doi.org/10.1007/s00300-017-2205-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-017-2205-1