Abstract
Background
The residual cancer burden class informs survival outcomes after neoadjuvant chemotherapy. We evaluated the prognostic ability of the RCB for survival outcomes in women with different phenotypic subtypes of breast cancer treated with neoadjuvant chemotherapy. Additional variables were assessed for inclusion with the RCB to further improve the model’s discriminative ability.
Patients and Methods
We conducted a retrospective review of patients completing at least 75% of the recommended cycles of neoadjuvant chemotherapy between 1 January 2010 and 31 December 2016. Phenotypic subtypes were defined by hormone receptor and human epidermal growth factor receptor 2 (HER2) status at diagnosis, classified as HR+/HER2−, HER2+, or triple-negative breast cancer (TNBC). The RCB class was calculated and survival endpoints of overall survival, recurrence-free survival, and distant recurrence-free survival were analyzed using Kaplan–Meier and Cox proportional hazards methods. The discriminative ability of the models was quantified by Harrell’s C-index.
Results
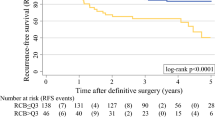
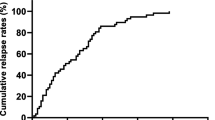
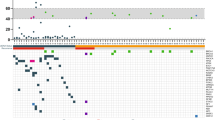
Overall, 532 women met the inclusion criteria. Median follow-up was 65 months. In univariate models, RCB was significantly associated with OS, RFS, and DRFS. The RCB class had good discriminative ability for OS, RFS, and DRFS survival, with Harrell’s C-indices of 0.68, 0.67, and 0.68, respectively. The RCB class discriminated well for each survival endpoint within HER2+ and TNBC, but did not discriminate well for HR+/HER2− (OS Harrell’s C-indices of 0.77, 0.75, and 0.52, respectively).
Conclusions
The RCB class was prognostic for OS, RFS, and DRFS after neoadjuvant chemotherapy, but prognostic discrimination between patients with subtype HR+/HER2− was not observed during the follow-up period for which the overall event rate was low.




Similar content being viewed by others
References
Spring L, Greenup R, Niemierko A, et al. Pathologic complete response after neoadjuvant chemotherapy and long-term outcomes among young women with breast cancer. J Natl Compr Canc Netw. 2017;15(10):1216–23.
Zhang P, Yin Y, Mo H, et al. Better pathologic complete response and relapse-free survival after carboplatin plus paclitaxel compared with epirubicin plus paclitaxel as neoadjuvant chemotherapy for locally advanced triple-negative breast cancer: a randomized phase 2 trial. Oncotarget. 2016;7(37):60647–56.
Li X, Yang J, Peng L, et al. Triple-negative breast cancer has worse overall survival and cause-specific survival than non-triple-negative breast cancer. Breast Cancer Res Treat. 2017;161(2):279–87.
Liedtke C, Mazouni C, Hess KR, et al. Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol. 2008;26(8):1275–2581.
Gupta G, Lee CD, Guye ML, et al. Unmet clinical need: Developing prognostic biomarkers and precision medicine to forecast early tumor relapse, detect chemo-resistance and improve overall survival in high-risk breast cancer. Ann Breast Cancer Ther. 2020;4(1):48–57.
Setiawan VW, Monroe KR, Wilkens LR, et al. Breast cancer risk factors defined by estrogen and progesterone receptor status: the multiethnic cohort study. Am J Epidemiol. 2009;169(10):1251–9.
Demonty G, Bernard-Marty C, Puglisi F, et al. Progress and new standards of care in the management of HER-2 positive breast cancer. Eur J Cancer. 2007;43(3):497–509.
Saw S, Lim J, Lim SH, et al. Patterns of relapse after neoadjuvant chemotherapy in breast cancer: implications for surveillance in clinical practice. Breast Cancer Res Treat. 2019;177(1):197–206.
Symmans WF, Peintinger F, Hatzis C, et al. Measurement of residual breast cancer burden to predict survival after neoadjuvant chemotherapy. J Clin Oncol. 2007;25(28):4414–22.
Symmans WF, Wei C, Gould R, Yu X, Zhang Y, et al. Long-term prognostic risk after neoadjuvant chemotherapy associated with residual cancer burden and breast cancer subtype. J Clin Oncol. 2017;35(10):1049–60.
Müller HD, Posch F, Suppan C, et al. Validation of residual cancer burden as prognostic factor for breast cancer patients after neoadjuvant therapy. Ann Surg Oncol. 2019;26(13):4274–83.
Giuliano AE, Edge SB, Hortobagyi GN. Eighth edition of the AJCC cancer staging manual: Breast cancer. Ann Surg Oncol. 2018;25(7):1783–5. https://doi.org/10.1245/s10434-018-6486-6.
MD Anderson Cancer Center. Residual Cancer Burden Calculator. MD Anderson Cancer Center website. Available at: http://www3.mdanderson.org/app/medcalc/index.cfm?pagename=jsconvert3. Accessed from 17 Jun 2021.
Campbell JI, Yau C, Krass P, et al. Comparison of residual cancer burden, American Joint Committee on Cancer staging and pathologic complete response in breast cancer after neoadjuvant chemotherapy: results from the I-SPY 1 TRIAL (CALGB 150007/150012; ACRIN 6657). Breast Cancer Res Treat. 2017;165(1):181–91.
Choi M, Park YH, Ahn JS, et al. Assessment of pathologic response and long-term outcome in locally advanced breast cancers after neoadjuvant chemotherapy: comparison of pathologic classification systems. Breast Cancer Res Treat. 2016;160(3):475–89.
Hamy AS, Darrigues L, Laas E, De Croze D, et al. Prognostic value of the Residual Cancer Burden index according to breast cancer subtype: Validation on a cohort of BC patients treated by neoadjuvant chemotherapy. PLoS One. 2020;15(6):e0234191.
Sheri A, Smith IE, Johnston SR, A’Hern R, et al. Residual proliferative cancer burden to predict long-term outcome following neoadjuvant chemotherapy. Ann Oncol. 2015;26(1):75–80.
Lee HJ, Park IA, Song IH, Kim SB, et al. Comparison of pathologic response evaluation systems after anthracycline with/without taxane-based neoadjuvant chemotherapy among different subtypes of breast cancers. PloS One. 2015;10(9):e0137885.
Howlader N, Cronin KA, Kurian AW, et al. Differences in breast cancer survival by molecular subtypes in the United States. Cancer Epidemiol Biomark Prev. 2018;27(6):619–26.
von Minckwitz G, Huang CS, Mano MS, Loibl S, et al. for the KATHERINE Investigators: trastuzumab emtansine for residual invasive HER2-positive breast cancer. N Engl J Med. 2019;380(7):617–28.
Masuda N, Lee SJ, Ohtani S, Im YH, et al. Adjuvant capecitabine for breast cancer after preoperative chemotherapy. N Engl J Med. 2017;376(22):2147–59.
Acknowledgments
The authors thank Geraldine M. Chadwick, AuD, who provided medical writing support on behalf of the Department of Surgery, Carolinas Medical Center.
Funding
None declared.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Erin A. Elder, Chad A. Livasy, Erin E. Donahue, Brittany Neelands, Alicia Patrick, Mckenzie Needham, Terry Sarantou, Lejla Hadzikadic-Gusic, Arielle L. Heeke, and Richard L. White Jr have declared no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Elder, E.A., Livasy, C.A., Donahue, E.E. et al. Residual Cancer Burden Class Associated with Survival Outcomes in Women with Different Phenotypic Subtypes of Breast Cancer After Neoadjuvant Chemotherapy. Ann Surg Oncol 29, 8060–8069 (2022). https://doi.org/10.1245/s10434-022-12300-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-12300-x