Abstract
The extensive use of nanoparticles under different industrial processes and their release into the environment are of major concerns in the present global scenario. In the present study, the effects of activated carbon-based TiO2 (AC-TiO2) nano-composite on the seed germination of Solanum lycopersicum (tomato) and Vigna radiata (mungbean) were investigated. The size of nanoparticles used in the study ranged from 30 to 50 nm, and their concentrations were from 0 to 500 mg L−1. The composites were synthesized by sol–gel method and further characterized by scanning electron microscopy, Energy-dispersive X-rays spectroscopy (EDX), Raman spectroscopy, Fourier transform infrared spectroscopy and X-ray diffraction to investigate all the surface structural and chemical properties of AC-TiO2 nano-composite. The results showed that increase in nano-composite concentration improves the germination rate and reduces germination time up to a certain concentration. Therefore, employing AC-TiO2 nano-composites in suitable concentration may promote the seed germination and also reduce the germination time in Solanum lycopersicum and Vigna radiata. Further, it may help to understand the interface of TiO2 nanoparticles with the environment and agriculture before its application to the field.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Nanotechnology is a rapidly growing and leading discipline of science and technology based industries, by playing a significant role in revolutionizing the agriculture field (Kumar et al. 2012; NAAS 2013; Srivastava et al. 2015a, b), economy, society and environment. It includes almost all branches of science and technology. The nanotechnology-based products are expected to be increased in the global market by US$ 1 trillion (Roco and Bainbridge 2001) including their synthesis processes (Maynard et al. 2006; Dawson 2008; Rejeski and Lekas 2008; Husen and Siddiqi 2014). As a consequence, it is generating both positive and negative responses from governments, scientists and social media throughout the world (Service 2000, 2003; Brumfiel 2003; Bai 2005; Yang et al. 2006). Due to its wide range of applications in various fields such as synthesis process, environmental remediation, cosmetic, agriculture sector, medicine and materials synthesis, it has been made a thrust area of research and development. However, its effects on the environment are mostly unexplored.
Nano-materials are found in both natural and engineered tailor forms. Engineered nano-materials have different physical and chemical properties as compared to the naturally occurring nanoparticles and thus have different effects on environment (Husen and Siddiqi 2014). USEPA (2010) categorized the different natural and engineered nano-materials into seven groups, viz. (1) carbonaceous (natural or engineered, e.g., fullerenes/buckyballs and nanotubes); (2) metal oxides [natural or engineered, e.g., titanium oxide (TiO2) and cerium oxide (CeO2)]; (3) zero-valent metals [engineered, e.g., nanoscale zero-valent iron (nZVI) and emulsified zero-valent iron (EZVI)]; (4) quantum dots [engineered, e.g., quantum dots made from cadmium selenide (CdSe) and cadmium telluride (CdTe)]; (5) dendrimers (engineered, e.g., hyperbranched polymers and dendrigraft polymers); (6) composite nano-polymers (engineered, e.g., made with two different nano-materials or resins); and (7) nano-silver (engineered, e.g., colloidal silver and polymeric silver).
Engineered nanoparticles has various applications in agricultural research, such as in reproductive science, transfer of agricultural and food waste to energy, nano-bioprocessing of enzymatic activity, disease prevention and other plant treatments using nanocides (Carmen et al. 2003). For instance, titanium dioxide (TiO2) is widely used in many process industries (including agrochemicals, cosmetic medicines, textiles, electronics, pharmaceutics and environmental remediation), due to its low cost, abundance, amphoteric nature and catalytic activities (Feizi et al. 2012). The release of nanoparticles in the environment through various routes including synthesis processes is hazardous from environmental point of view. Therefore, proper recycling and disposal of nano-based products are imperative. Further, the disposal of nano-materials after use in the agricultural systems and understanding of its effect on the same are of vital importance.
Generally, the effects of nano-materials on plant and soil depend upon their particle size and nature of crystalline structure (Feizi et al. 2012, 2013; Clément et al. 2013; Dehkourdi and Mosavi 2013; Song et al. 2013b). For example, the effect of nano-sized TiO2 on various plants’ seed germination and growth showed ambiguous results, because of its positive and negative effects ranging from strong toxicity to the root–shoot systems to growth-stimulating effects (Song et al. 2013b). Various positive and negative effects of TiO2 nanoparticle associated with the seed germination and plant growth are summarized in Table 1. However, various researches found the negative effects of TiO2 nanoparticles on a few plant species (García et al. 2011; Mushtaq 2011; Jośko and Oleszczuk 2013; Song et al. 2013a). Moreover, various other effects of nano-materials on the plants and soil ecology interaction are still unknown (Lin and Xing 2007). However, other studies supported that TiO2 application to soil enhances the chlorophyll content and enzymatic activities such as peroxidase, catalase and nitrate reductase which positively affects on the growth and production by improving essential element content in plant tissue in various crops (Hruby et al. 2002; Feizi et al. 2012). The use of TiO2 nanoparticles may possibly be a new approach to overcome problems with seed germination in some plant species, particularly medicinal plants which have lower germination rate dormancy period (Feizi et al. 2013).
Understanding of the degree at which nanoparticles affect seed germination and plant development is an important issue. It depends upon the concentration, nature and size of nanoparticles. This could also have economic significance for agriculture (Ju-Nam and Lead 2008; Gong et al. 2011; Gottschalk and Nowack 2011; Jośko and Oleszczuk 2013). Presently, carbon-based TiO2 nano-materials (Mattle and Thampi 2013), carbon nano-tubes (Ouyang et al. 2013), Fe2O3–TiO2 composite (Ouyang et al. 2013) and N-doped TiO2 (Sun et al. 2008) catalysts are widely used for the remediation of various environmental contaminations. During processing, a few amounts of these composites are released in the environment and reached the agriculture systems through various processes. However, the environmental impacts of TiO2 and other such semiconductors used as catalysts have not been studied extensively. Therefore, in the present study, effect of TiO2 and activated carbon nano-composite (AC-TiO2) on the seed germination of two important agricultural crops, viz. Solanum lycopersicum and Vigna radiata, is studied. To check out its effect on agricultural system, this work aims to achieve following objectives dealing with synthesis, characterization and germination effects as:
-
1.
Synthesis of activated carbon-based TiO2 (AC-TiO2) nano-composite through sol–gel method and its characterization using various instrumentation techniques and
-
2.
Evaluation of effect nano-composite on the seed germination, root and shoot length and weight of two important agricultural crops
2 Materials and methods
2.1 Chemicals
Titanium tetraisopropoxide (TTIP) and activated carbon (<20 μm) were procured from Sigma-Aldrich. Distilled water was used for all the synthesis processes and for the preparation of nanoparticle suspension.
2.2 Preparation of AC-TiO2 nano-composite
Activated carbon-based titanium dioxide (AC-TiO2) nano-composite was synthesized using the sol–gel method by using titanium tetraisopropoxide (97 %) as a binder and commercially available activated carbon (Inoue et al. 1994; Horie et al. 1998; Singh et al. 2015). During the preparation process, 35.8 g of TTIP (97 %) was dissolved in 180 mL of 99.9 % propanol and 20 mL of 34 % HCl (w/v) and sonicated for 1 h (h) for homogenization. The resulting solution was diluted to 1000 mL by adjusting pH (pH = 3) with NaOH. Ten grams of activated carbon and 10 g of P25 TiO2 particles were mixed together and stirred both on magnetic and mechanical stirrer for 3 h in the presence of the above TTIP, HCl and propanol solution. Obtained gel solution was then filtered through membrane filter (0.45 μm) and oven-dried at 80 °C for 24 h. The dried samples were crushed and calcinated at 350 °C temperature for 3 h as suggested by Kubo et al. (2007).
2.3 Characterization of nano-composite
Various characterization techniques were used to study and analyze the nature, surface and structural properties and phase composition of the nano-composite used. Surface characteristics were observed using scanning electron microscopy (FEI QUANTUM 600F). Phase composition of samples being used, their crystalline nature and size were determined using X-ray diffraction (XRD) carried out in a SIEMENS D500 with Cu-Kα radiation in region varying from 2θ = 10°–80°. The functional groups of the composite material were analyzed by Fourier transform infrared (FTIR) spectroscopy (Thermo NICOLET 5700, USA) spectra of samples (with KBr) in the spectral range of 4000–500 cm−1. Raman spectra of samples were obtained using Jobin–Yvon Horiba HR800 Raman spectrometer.
2.4 Preparation of composite suspensions
The AC-TiO2 nano-composites were suspended directly in deionized water and dispersed by ultrasonic vibration for 30 min. Small magnetic bars were placed in the suspensions for stirring before use to avoid aggregation of the particles.
2.5 Seeds
Seeds of important agricultural crops (viz. Vigna radiata and Solanum lycopersicum) were collected from the Institute of Agricultural Science and Technology, Banaras Hindu University, Varanasi, India.
2.6 Seed germination and exposure
The collected seeds were washed in tap water initially, immersed in a 10 % sodium hypochlorite solution for 10 min and then rinsed three times with deionized water to ensure surface sterility. Then, the seeds were soaked in nano-composite suspensions solution for about 12 h. One piece of filter paper was put into each 100-mm petri dish, and 5 mL of a test medium was added. Seeds were then transferred onto the filter paper, with 10 seeds per dish with about 1 cm distance between seeds (Yang and Watts 2005). Petri dishes were covered and sealed with paraffin and placed in the dark in a growth chamber at 25 °C. After 24 h, the germination of seeds was checked, on regular basis. The root length and shoot length were measured after 3 days when germination was halted, and seedling root length was measured by a millimeter ruler to observe the different effects between seed soaking and incubation process on the root and shoot elongation.
3 Results and discussion
3.1 Scanning electron microscopy
Surface morphology of composite was characterized by SEM as shown in (Fig. 1). The rough porous surface of activated carbon is resulted from the growth of TiO2 nanoparticles on it. Most of the TiO2 nanoparticles were entirely filled into interstitial pores of activated carbon particles (Fig. 1a, b). Presence of TiO2 along with activated carbon has been also inferred by EDX analysis results shown in Fig. 1c. This revealed that the TiO2 nanoparticles get adsorbed on the surface of activated carbon to form AC-TiO2 nano-composites.
3.2 Raman spectra analysis
The Raman spectra of activated carbon, bare TiO2 nanoparticles and TiO2 adsorbed on the surface of activated carbon (AC-TiO2 nano-composite) are shown in Fig. 2. As per the observation, spectra of activated carbon (Fig. 2a) exhibit two well-resolved bands, D (1332 cm−1) and G (1585 cm−1), which clearly indicates vibration in C–C bond in activated carbon (Cuesta et al. 1994). Further, Fig. 2b showed the Raman spectra of bare TiO2 with each unit cell possessing tetragonal structure (Hyun et al. 2005). Raman spectra of single crystal indicate the four modes which appear at 133, 190, 386, 506 and 630 cm−1 (Šćepanović et al. 2009). The first peak appearing at 133 cm−1 is slightly broader and shifted than that of a bulk TiO2 crystal (Zhang et al. 2000). As compared to short-range order of anatase phase in weak broader phase in high-frequency region, certain degree of long-range order exists at 133 cm−1 peak (Arora et al. 2007). Figure 2c showed the Raman spectra of AC-TiO2 nano-composites having peaks at same wave numbers as that of spectra of TiO2. The variation in intensity at given described peaks reflected that activated carbon is successfully exfoliated and incorporated in the nano-composite.
3.3 Fourier transform infrared (FTIR) analysis
FTIR spectra of AC-TiO2 nanoparticles showed different peaks at different wave numbers revealing various absorption patterns (Fig. 3). Absorption peak at 3415 cm−1 represents the stretching of hydroxyl (–OH) group in water as moisture (Ba-Abbad et al. 2012), whereas peak at 1632 cm−1 shows the stretching of titanium carboxylate, which was the product of TTIP and ethanol used in sol–gel method. Further, absorption peak at 757 cm−1 represents the stretching of Ti–O bond which is the characteristic attribute of the formation of TiO2 nanoparticles (Hema et al. 2013). FTIR study of AC-TiO2 nano-composite shows the shift in the O–H vibration band toward lower wave number (3400 cm−1). These shifts confirm the alteration of acid–base characteristics of –OH group in the used samples. Further, the bands near 600 cm−1 are assigned to the stretch vibration of Ti–O bond and prove that the TiO2 particle is well distributed on the surface of activated carbon (Zhang et al. 2012).
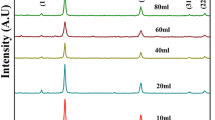
3.4 X-ray diffraction of nano-composite
X-ray diffraction analysis was performed to assay the phase composition, crystalline nature and size of prepared AC-TiO2 nano-composites (Fig. 4). Various diffraction peaks (Fig. 4) at 2θ = 25.40°, 48.02°, 54.19° and 62.72° were given by AC-TiO2 nano-composite, which were assigned to (101), (200), (105) and (103) reflections of anatase phase and peaks at 2θ = 27.48°, 36.07°, 37.80° and 69.00° being assigned to (001), (021), (210) and (220) reflect the rutile phase of TiO2. The average intensity of rutile phase is considerably less as compared to that of anatase phase. Average crystalline size can be determined using Scherrer’s equation (Borchert et al. 2005) as:
where K = Scherer constant, λ = X-ray wavelength, β = the peak width of half maximum, and θ = Bragg diffraction angle.
3.5 Effect of AC-TiO2 nano-composite on seed germination
To test the effect of AC-TiO2 nano-composite on seed germination and seedling growth, the seeds of V. radiata (Mung) and S. lycopersicum (Tomato) were placed in petri dish and treated with 5 mL of AC-TiO2/water mixture of different concentrations. The seeds without AC-TiO2 suspension treatment were used for control experiments (Fig. 5). After 3 days of incubation, AC-TiO2 was found to accelerate the process of seed germination and significantly shortened the germination time as compared to the control one. The root-to-shoot lengths of AC-TiO2-treated seed as shown in Fig. 5 were also found much longer than that of the control seeds. Moreover, the root length of V. radiata showed higher value at the lower concentration of AC-TiO2; however, the overall root length decrease with further increase in the concentration.
Figure 6 shows that germination of lower-concentration-treated S. lycopersicum seeds was more than that of control seeds on the second day. During the next few days, the germination rate was dramatically faster for seeds treated with AC-TiO2 than that of the control seeds. The germination percentage for the control seeds averaged 80 % in 4 days, while germination percentage of the AC-TiO2-treated seeds averaged 100 % (higher concentration) in 4 days, 100 % (medium concentration) in 4 days and 90 % in lower concentration (Fig. 6). It indicates that the accelerated seed germination could be caused by increasing concentration of nano-composite, which might be due to the penetration of the seed husks by TiO2. As illustrated in Fig. 6, at stage I, the AC-TiO2 was found to be densely deposited on the seeds’ surface and penetrated seed husks which supports and allows water uptake inside the seeds. In case of S. lycopersicum, the germination rate was 0 % in 4 days in control, 10 % in lower concentration, 35 % in medium concentration and 80 % in higher concentration. After 8 days, the germination rate was increased by 0–50 % in control, 50 % in lower concentration, 95 % in medium concentration and 90 % in higher concentration. Zhang et al. (2015) demonstrated that the nano-sized TiO2 helps the water absorption by the Spinach seed which helps in the enhancement of the germination rate. Khodakovskaya et al. (2009) also supported the same in case of carbon nanotube. Water uptake is an important process in the seed germination as mature seeds are fairly dry and need water to initiate the cellular metabolisms and growth.
Generally, these nanoparticles enhance the germination rate in various plant species. Mature seeds are relatively dry and need uptake of significant amounts of water to start their cellular metabolism and growth resume. Nanoparticles may create new pores in the seed coat and therefore facilitate the process of water uptake inside the seed embryo, and therefore, germination rate. Various other studies reported that TiO2 in the nano-range promotes the seed germination, photosynthetic activity and nitrogen metabolisms which cumulatively promotes the growth of plant species at a suitable range of concentrations (Zheng et al. 2005; Hong et al. 2005; Yang et al. 2007). Further, it increases the activity of several enzymes and promotes the adsorption of nitrate, accelerating the transformation of inorganic into organic nitrogen. However, normal-sized TiO2 does not have such effects (Ma et al. 2010a, b).
AC-TiO2 composite had positive impacts on the germination of V. radiata seeds, which can be attributed to the fact that TiO2 was able to penetrate the seed husks (Fig. 7). The penetration might break the husks to facilitate water uptake, resulting in rapid seed germination and higher percentage of germination rates. However, at the stage of seedling growth, activated carbon may be providing moisture to the seed. Furthermore, stems and roots of the higher-concentration-treated seedlings were longer than those of the control. Zhang et al. (2015) found that in the case of graphene, it penetrates the seed husk which might break the husk to facilitate water uptake, resulting in faster germination as well as higher germination rate. Carbon nano-materials are known for their light weight and extreme conductivity which occupy a unique place in agriculture because of their abilities to enhance the seedling growth and development. Effect of nano-materials on the plant growth also depends upon the type of nano-materials, size-specific area, functional groups, concentration, plant species, soil type and condition (Lin and Xing 2007; Ge et al. 2011; Rico et al. 2011; Josko and Oleszczuk 2013).
Seed germination was higher in lower and medium concentrations of AC-TiO2, whereas root-to-shoot ratio was found higher in the lower concentration. It showed that the concentration of nano-composite plays a vital role in the plant growth and development. The increase in the seed germination might be due to the photo-generation of active oxygen like superoxide and hydroxide anions which causes re-activation of aged seed. Activated carbon present in the nano-composite provides large surface area and moisture for seed germination. It also enhances the penetrability of seed capsule, facilitating the admission of water and dioxygen into the cell, thereby resulting in an increase in the seed germination. TiO2 also induces oxidation–reduction via free superoxide radical during the germination. The oxygen produced in such processes could be used for respiration which further promotes the seed germination (Zheng et al. 2005).
4 Conclusions
The release of nano-materials into the environment affects various plant growth mechanisms and development from the seed germination to pollination. We observed that the activated carbon-based TiO2 (AC-TiO2) nano-composite had positive impacts on the germination of Vigna radiata and Solanum lycopersicum seeds, which can be attributed to the fact that TiO2 was able to penetrate the seed husks. It envisaged that the elongation in root-to-shoot ratio may be related to the concentration of catalysts, but further studies are needed at cellular and molecular level for the best understanding of the mechanisms of reaction of catalysts with seed. The dose of the nano-composites (AC-TiO2) was studied on the germination of seed and by measuring their root and shoot growth. The results indicated that AC-TiO2 nano-composite enhanced the seed germination in plants and also enhanced the shoot-to-root ratio depending on the concentration of materials. At increased concentration, the germination is enhanced, but the growth of root and shoot was either decreased or remained stable as compared to the control. Overall, these results will be helpful for further understanding of the mechanisms of interaction of nano-materials with the plants species.
References
Arora AK, Rajalakshmi M, Ravindran TR, Sivasurbramanian V (2007) Raman spectroscopy of optical phonon confinement in nanostructured materials. J Raman Spectrosc 38:604–617
Ba-Abbad MM, Kadhum AAH, Mohamad AB, Takriff MS, Sopian K (2012) Synthesis and catalytic activity of TiO2 nanoparticles for photochemical oxidation of concentrated chlorophenols under direct solar radiation. Int J Electrochem Sci 7:4871–4888
Bai CL (2005) Ascent of nanoscience in China. Science 309:61–63
Borchert H, Shevchenko EV, Robert A, Mekis I, Kornowski A, Grubel G, Weller H (2005) Determination of nanocrystal sizes: a comparison of TEM, SAXS, and XRD studies of highly monodisperse CoPt3 particles. Langmuir 21:1931–1936
Brumfiel G (2003) Nanotechnology: a little knowledge. Nature 424:246–248
Carmen IU, Chithra P, Huang Q, Takhistov P, Liu S, Kokini JL (2003) Nanotechnology: a new frontier in food science. Food Technol 57:24–29
Castiglione MR, Giorgetti L, Geri C, Cremonini R (2011) The effects of nano-TiO2 on seed germination, development and mitosis of root tip cells of Vicia narbonensis L. and Zea mays L. J Nanopart Res 13(6):2443–2449
Clément L, Hurel C, Marmier N (2013) Toxicity of TiO2 nanoparticles to cladocerans, algae, rotifers and plants—effects of size and crystalline structure. Chemosphere 90:1083–1090
Cuesta P, Dhamelincourt J, Laureyns A, Martinez-Alonso JM, Tasc D (1994) Raman microprobe studies on carbon materials. Carbon 32:523–1532
Dawson NG (2008) Sweating the small stuff, environmental risk and nanotechnology. BioSci 58:690
Dehkourdi EH, Mosavi M (2013) Effect of anatase nanoparticles (TiO2) on parsley seed germination (Petroselinum crispum) in vitro. Biol Trace Elem Res 155:283–286
Feizi H, Moghaddam PR, Shahtahmassebi N, Fotovat A (2012) Impact of bulk and nanosized titanium dioxide (TiO2) on wheat seed germination and seedling growth. Biol Trace Elem Res 146:101–106
Feizi H, Kamali M, Jafari L, Moghaddam PR (2013) Phytotoxicity and stimulatory impacts of nanosized and bulk titanium dioxide on fennel (Foeniculum vulgare Mill). Chemosphere 91:506–511
García A, Espinosa R, Delgado L, Casals E, Gonzalez E, Puntes V, Barata C, Font X, Sanchez A (2011) Acute toxicity of cerium oxide, titanium oxide and iron oxide nanoparticles using standardized tests. Desalination 269:136–141
Ge C, Du J, Zhao L, Wang L, Liu Y, Li D, Yang Y, Zhou R, Zhao Y, Chai Z, Chen C (2011) Binding of blood proteins to carbon nanotubes reduces cytotoxicity. Proc Natl Acad Sci 108(41):16968–16973
Gong N, Shao K, Feng W, Lin Z, Liang C, Sun Y (2011) Biotoxicity of nickel oxide nanoparticles and bio-remediation by microalgae Chlorella vulgaris. Chemosphere 83(4):510–516
Gottschalk F, Nowack B (2011) The release of engineered nanomaterials to the environment. J Environ Monit 13:1145–1155
Haghighi M, da Silva JAT (2014) The effect of N-TiO2 on tomato, onion, and radish seed germination. J Crop Sci Biotechnol 17(4):221–227
Hema M, Arasi AY, Tamilselvi P, Anbarasan R (2013) Titania nanoparticles synthesized by sol–gel technique. Chem Sci Trans 2:239–245
Hong F, Zhou J, Liu C, Yang F, Wu C, Zheng L, Yang P (2005) Effects of nano-TiO2 on photochemical reaction of chloroplasts of spinach. Biol Trace Elem Res 105:269–279
Horie Y, Taya M, Tone S (1998) Effect of cell adsorption on photosterilization of Escherichia coli over titanium dioxide-activated charcoal granules. J Chem Eng 31:922–929
Hruby M, Cigler P, Kuzel S (2002) Contribution to understanding the mechanism of titanium action in plant. J Plant Nutr 25:577–598
Husen A, Siddiqi KS (2014) Phytosynthesis of nanoparticles: concept, controversy and application. Nanoscale Res Lett 9:1–24
Hyun CC, Young MJ, Seung BK (2005) Size effects in the Raman spectra of TiO2 nanoparticles. Vib Spectrosc 37:33–38
Inoue H, Matsuyama T, Liu BJ, Sakata T, Mori H, Yoneyama H (1994) Photocatalytic activities for carbon dioxide reduction of TiO2 microcrystals prepared in SiO2 matrices using a sol–gel method. Chem Lett 3:653–656
Jośko I, Oleszczuk P (2013) Influence of soil type and environmental conditions on ZnO, TiO2 and Ni nanoparticles phytotoxicity. Chemosphere 92:91–99
Ju-Nam Y, Lead JR (2008) Manufactured nanoparticles: an overview of their chemistry, interactions and potential environmental implications. Sci Total Environ 400(1):396–414
Khodakovskaya M, Dervishi E, Mahmood M, Xu Y, Li Z, Watanabe F, Biris AS (2009) Carbon nanotubes are able to penetrate plant seed coat and dramatically affect seed germination and plant growth (retracted article. see, vol. 6, pp. 7541, 2012). ACS Nano 3:3221–3227
Kubo M, Fukuda H, Chua XJ, Yonemoto T (2007) Kinetics of ultrasonic degradation of phenol in the presence of composite particles of titanium dioxide and activated carbon. Ind Eng Chem Res 46:699–704
Kumar R, Roopan SM, Prabhakarn A, Khanna VG, Chakroborty S (2012) Agricultural waste Annona squamosa peel extract: biosynthesis of silver nanoparticles. Spectro Acta A Mol Biomol Spectrosc 90:173–176
Lin D, Xing B (2007) Phytotoxicity of nanoparticles: inhibition of seed germination and root growth. Environ Pollut 150:243–250
Ma X, Geiser-Lee J, Deng Y, Kolmakov A (2010a) Interactions between engineered nanoparticles (ENPs) and plants: phytotoxicity, uptake and accumulation. Sci Total Environ 408:3053–3061
Ma Y, Kuang L, He X, Bai W, Ding Y, Zhang Z, Zhao Y, Chai Z (2010b) Effects of rare earth oxide nanoparticles on root elongation of plants. Chemosphere 78:273–279
Mattle MJ, Thampi KR (2013) Photocatalytic degradation of Remazol Brilliant Blue® by sol–gel derived carbon-doped TiO2. Appl Catal B Environ 140–141:348–355
Maynard AD, Aitken RJ, Butz T, Colvin V, Donaldson K, Oberdörster G, Philbert MA, Ryan J, Seaton A, Stone V, Tinkle SS, Tran L, Walker NJ, Warheit DB (2006) Safe handling of nanotechnology. Nature 444:267–269
Mushtaq YK (2011) Effect of nanoscale Fe3O4, TiO2 and carbon particles on cucumber seed germination. J Environ Sci Health Part A Tox Hazard Subst Environ Eng 46:1732–1735
NAAS (2013) Nanotechnology in agriculture: Scope and Current Relevance. Policy paper no. 63. National Academy of Agricultural Sciences, New Delhi
Ouyang K, Xie S, Ma XO (2013) Photocatalytic activity of TiO2 supported on multi-walled carbon nanotubes under simulated solar irradiation. Ceram Int 39:7531–7536
Rejeski D, Lekas D (2008) Nanotechnology field observations: scouting the new industrial west. J Clean Prod 16:1014–1017
Rico CM, Majumdar S, Duarte-Gardea M, Peralta-Videa JR, Gardea-Torresdey JL (2011) Interaction of nanoparticles with edible plants and their possible implications in the food chain. J Agric Food Chem 59:3485–3498
Roco MC, Bainbridge WS (2001) Societal implications of nanoscience and nanotechnology. Kluwer, Boston, pp 3–4
Šćepanović MJ, Grujić-Brojčin M, Dohčević-Mitrović ZD, Popović ZV (2009) Characterization of anatase TiO2 nanopowder by variable-temperature raman spectroscopy. Sci Sinter 41:67–73
Service RF (2000) Is nanotechnology dangerous. Science 290:1526–1527
Service RF (2003) Nanomaterials show signs of toxicity. Science 300:243
Singh P, Vishnu MC, Sharma KK, Singh R, Madhav S, Tiwary D, Mishra PK (2015) Comparative study of dye degradation using TiO2-activated carbon nanocomposites as catalysts in photocatalytic, sonocatalytic and photosonocatalytic reactor. Desalin Water Treat. doi:10.1080/19443994.2015.1108871
Song U, Jun H, Waldman B, Roh J, Kim Y, Yi J, Lee EJ (2013a) Functional analyses of nanoparticle toxicity: a comparative study of the effects of TiO2 and Ag on tomatoes (Lycopersicon esculentum). Ecotox Environ Saf 93:60–67
Song U, Shin M, Lee G, Roh J, Kim Y, Lee EJ (2013b) Functional analysis of TiO2 nanoparticle toxicity in three plant species. Biol Trace Elem Res 155:93–103
Srivastava N, Singh J, Srivastava M, Ramteke PW, Mishra PK (2015a) Improved production of reducing sugars from rice straw using crude cellulase activated with Fe3O4/Alginate nanocomposite. Bioresour Technol 183:262–266
Srivastava N, Srivastava M, Mishra PK, Singh P, Ramteke PW (2015b) Application of cellulases in biofuels industries: an overview. J Biofuels Bioenergy 1(1):55–63
Sun J, Qiao L, Sun S, Wang G (2008) Photocatalytic degradation of Orange G on nitrogen-doped TiO2 catalysts under visible light and sunlight irradiation. J Hazard Mater 155:312–319
U.S. Environmental Protection Agency (2010) Emerging contaminants-nanoparticles. www3.epa.gov/region9/mediacenter/nano-ucla/emerging-contaminant-nanomaterials.pdf. Accessed 20 Dec 2015
Yang L, Watts DJ (2005) Particle surface characteristics may play an important role in phytotoxicity of alumina nanoparticles. Toxicol Lett 158(2):122–132
Yang K, Wang XL, Zhu LZ, Xing BS (2006) Competitive sorption of pyrene, phenanthrene, and naphthalene on multiwalled carbon nanotubes. Environ Sci Technol 40:5804–5810
Yang F, Liu C, Gao F, Su M, Wu X, Zheng L, Hong F, Yang P (2007) The improvement of spinach growth by nano-anatase TiO2 treatment is related to nitrogen photoreduction. Biol Trace Elem Res 119:77–88
Zhang WF, Yl He, Zhang MS, Yin Z (2000) Raman scattering study on anatase TiO2 nanocrystals. J Phys D Appl Phys 33:912
Zhang Z, Xu Y, Ma X, Li F, Liu D, Chen Z, Dionysiou DD (2012) Microwave degradation of methyl orange dye in aqueous solution in the presence of nano-TiO2-supported activated carbon (supported-TiO2/AC/MW). J Hazard Mater 209:271–277
Zhang M, Gao B, Chen J, Li Y (2015) Effects of graphene on seed germination and seedling growth. J Nanopart Res 17:1–8
Zheng L, Hong F, Lu S, Liu C (2005) Effect of nano-TiO2 on strength of naturally aged seeds and growth of spinach. Biol Trace Elem Res 104(1):83–91
Acknowledgments
Authors gratefully acknowledge the financial support provided by the University Grants Commission (UGC) and Council for Scientific & Industrial Research (CSIR), New Delhi, for providing Senior Research Fellowships. N.S. thankfully acknowledge to DST, New Delhi, India for providing the Women scientist-B fellowship (SEED/DISHA/WOSB/047/2012/G). PS is also highly thankful to Central Instrument Facility Center, IIT-BHU for providing instrumentation facility. Authors are also gratefully acknowledge Prof. S.P. Singh, IAS-BHU, for providing seed and Dr. R.K. Singh, Lab-in-charge, RAMAN Spectroscopy Laboratory, Department of Physics, BHU, Varanasi for providing support in RAMAN spectroscopy. Annonymous reviewers and the Editors are also gratefully acknowledged for their critics and comments for improving the present article.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Singh, P., Singh, R., Borthakur, A. et al. Effect of nanoscale TiO2-activated carbon composite on Solanum lycopersicum (L.) and Vigna radiata (L.) seeds germination. Energ. Ecol. Environ. 1, 131–140 (2016). https://doi.org/10.1007/s40974-016-0009-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40974-016-0009-8