Abstract
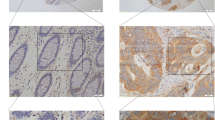
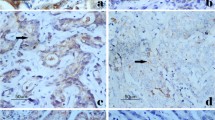
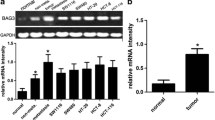
BRCA1-associated protein 1 (BAP1) has been reported as a novel tumor suppressor, while in gastric adenocarcinoma, the function of this protein was still await to be uncovered. Based on a large group of patients with gastric adenocarcinoma, our study aimed to have a further understanding about the correlation of BAP1 expression and patients’ clinical outcomes. We performed quantitative PCR and Western blot to examine BAP1 expression in 38 cases of gastric adenocarcinoma samples and adjacent non-cancerous tissues. Immunochemistry was used to evaluate BAP1 expression in a large cohort of 474 paraffin-embedded specimens. The clinical and prognostic significance of BAP1 expression was statistically analyzed. Postoperative survival between groups was using Kaplan-Meier analysis. BAP1 was overexpressed in paracancerous normal mucosa compared with gastric cancer. Decreased BAP1 expression was associated with higher histologic grade (P = 0.044), tumor infiltration (P < 0.001), metastasis status (P = 0.023), and TNM stage (P < 0.001). Patients with low expression of BAP1 had shorter overall survival compared with those with high expression (P < 0.001). Patients’ survival in stage N0 could be stratified by the expression of BAP1. Multivariate analysis showed that in gastric adenocarcinoma, BAP1 expressing level was an independent prognostic factor (RR = 0.575, P < 0.001). Decreased expression of BAP1 suggests pessimistic prognosis for gastric adenocarcinoma patients. Further studies are warranted.




Similar content being viewed by others
Abbreviations
- BAP1:
-
BRCA1-associated protein 1
- TNM:
-
Tumor-node-metastasis
- IHC:
-
Immunohistochemistry
- RT-PCR:
-
Reverse transcription polymerase chain reaction
- WB:
-
Western blot
References
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30.
Macdonald JS, Smalley SR, Benedetti J, Hundahl SA, Estes NC, Stemmermann GN, et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med. 2001;345:725–30.
Gulubova M, Manolova I, Kyurkchiev D, Julianov A, Altunkova I. Decrease in intrahepatic cd56+ lymphocytes in gastric and colorectal cancer patients with liver metastases. APMIS. 2009;117:870–9.
Iwasa S, Yanagawa T, Fan J, Katoh R. Expression of cxcr4 and its ligand sdf-1 in intestinal-type gastric cancer is associated with lymph node and liver metastasis. Anticancer Res. 2009;29:4751–8.
Mrena J, Wiksten JP, Kokkola A, Nordling S, Ristimaki A, Haglund C. Cox-2 is associated with proliferation and apoptosis markers and serves as an independent prognostic factor in gastric cancer. Tumour Biol. 2010;31:1–7.
Tamura Y, Higashi M, Kitamoto S, Yokoyama S, Osako M, Horinouchi M, et al. Muc4 and muc1 expression in adenocarcinoma of the stomach correlates with vessel invasion and lymph node metastasis: an immunohistochemical study of early gastric cancer. PLoS One. 2012;7, e49251.
Shah MA, Khanin R, Tang L, Janjigian YY, Klimstra DS, Gerdes H, et al. Molecular classification of gastric cancer: a new paradigm. Clin Cancer Res. 2011;17:2693–701.
Song IS, Ha GH, Kim JM, Jeong SY, Lee HC, Kim YS, et al. Human znf312b oncogene is regulated by sp1 binding to its promoter region through DNA demethylation and histone acetylation in gastric cancer. Int J Cancer. 2011;129:2124–33.
Wang B, Matsuoka S, Ballif BA, Zhang D, Smogorzewska A, Gygi SP, et al. Abraxas and rap80 form a brca1 protein complex required for the DNA damage response. Science. 2007;316:1194–8.
Wang K, Kan J, Yuen ST, Shi ST, Chu KM, Law S, et al. Exome sequencing identifies frequent mutation of arid1a in molecular subtypes of gastric cancer. Nat Genet. 2011;43:1219–23.
Bott M, Brevet M, Taylor BS, Shimizu S, Ito T, Wang L, et al. The nuclear deubiquitinase bap1 is commonly inactivated by somatic mutations and 3p21.1 losses in malignant pleural mesothelioma. Nat Genet. 2011;43:668–72.
Harbour JW, Onken MD, Roberson ED, Duan S, Cao L, Worley LA, et al. Frequent mutation of bap1 in metastasizing uveal melanomas. Science. 2010;330:1410–3.
Jensen DE, Proctor M, Marquis ST, Gardner HP, Ha SI, Chodosh LA, et al. Bap1: a novel ubiquitin hydrolase which binds to the brca1 ring finger and enhances brca1-mediated cell growth suppression. Oncogene. 1998;16:1097–112.
Machida YJ, Machida Y, Vashisht AA, Wohlschlegel JA, Dutta A. The deubiquitinating enzyme bap1 regulates cell growth via interaction with hcf-1. J Biol Chem. 2009;284:34179–88.
Scheuermann JC, de Ayala Alonso AG, Oktaba K, Ly-Hartig N, McGinty RK, Fraterman S, et al. Histone h2a deubiquitinase activity of the polycomb repressive complex pr-dub. Nature. 2010;465:243–7.
Stokes MP, Rush J, Macneill J, Ren JM, Sprott K, Nardone J, et al. Profiling of uv-induced atm/atr signaling pathways. Proc Natl Acad Sci U S A. 2007;104:19855–60.
Ventii KH, Devi NS, Friedrich KL, Chernova TA, Tighiouart M, Van Meir EG, et al. Brca1-associated protein-1 is a tumor suppressor that requires deubiquitinating activity and nuclear localization. Cancer Res. 2008;68:6953–62.
Guenard F, Labrie Y, Ouellette G, Beauparlant CJ, Durocher F, BRCAs I. Genetic sequence variations of brca1-interacting genes aurka, bap1, bard1 and dhx9 in french canadian families with high risk of breast cancer. J Hum Genet 2009;54:152–161.
Coupier I, Cousin PY, Hughes D, Legoix-Ne P, Trehin A, Sinilnikova OM, et al. Bap1 and breast cancer risk. Fam Cancer. 2005;4:273–7.
Pena-Llopis S, Vega-Rubin-de-Celis S, Liao A, Leng N, Pavia-Jimenez A, Wang S, et al. Bap1 loss defines a new class of renal cell carcinoma. Nat Genet. 2012;44:751–9.
Testa JR, Cheung M, Pei J, Below JE, Tan Y, Sementino E, et al. Germline bap1 mutations predispose to malignant mesothelioma. Nat Genet. 2011;43:1022–5.
Wang W, Lv L, Pan K, Zhang Y, Zhao JJ, Chen JG, et al. Reduced expression of transcription factor ap-2alpha is associated with gastric adenocarcinoma prognosis. PLoS One. 2011;6, e24897.
He F, Wu HN, Cai MY, Li CP, Zhang X, Wan Q, et al. Inhibition of ovarian cancer cell proliferation by pien tze huang via the akt-mtor pathway. Oncol Lett. 2014;7:2047–52.
Fan LH, Tang LN, Yue L, Yang Y, Gao ZL, Shen Z. Bap1 is a good prognostic factor in advanced non-small cell lung cancer. Clin Invest Med. 2012;35:E182–189.
Tang J, Xi S, Wang G, Wang B, Yan S, Wu Y, et al. Prognostic significance of brca1-associated protein 1 in colorectal cancer. Med Oncol. 2013;30:541.
Angeloni D. Molecular analysis of deletions in human chromosome 3p21 and the role of resident cancer genes in disease. Brief Funct Genomic Proteomic. 2007;6:19–39.
Tyagi S, Chabes AL, Wysocka J, Herr W. E2f activation of s phase promoters via association with hcf-1 and the mll family of histone h3k4 methyltransferases. Mol Cell. 2007;27:107–19.
Dey A, Seshasayee D, Noubade R, French DM, Liu J, Chaurushiya MS, et al. Loss of the tumor suppressor bap1 causes myeloid transformation. Science. 2012;337:1541–6.
Misaghi S, Ottosen S, Izrael-Tomasevic A, Arnott D, Lamkanfi M, Lee J, et al. Association of c-terminal ubiquitin hydrolase brca1-associated protein 1 with cell cycle regulator host cell factor 1. Mol Cell Biol. 2009;29:2181–92.
Sowa ME, Bennett EJ, Gygi SP, Harper JW. Defining the human deubiquitinating enzyme interaction landscape. Cell. 2009;138:389–403.
Yu H, Mashtalir N, Daou S, Hammond-Martel I, Ross J, Sui G, et al. The ubiquitin carboxyl hydrolase bap1 forms a ternary complex with yy1 and hcf-1 and is a critical regulator of gene expression. Mol Cell Biol. 2010;30:5071–85.
Alatengbaolide, Lin D, Li Y, Xu H, Chen J, Wang B, et al. Lymph node ratio is an independent prognostic factor in gastric cancer after curative resection (r0) regardless of the examined number of lymph nodes. Am J Clin Oncol. 2013;36:325–30.
Pacelli F, Cusumano G, Rosa F, Marrelli D, Dicosmo M, Cipollari C, et al. Multivisceral resection for locally advanced gastric cancer: an Italian multicenter observational study. JAMA Surg. 2013;148:353–60.
Gyorffy B, Surowiak P, Budczies J, Lanczky A. Online survival analysis software to assess the prognostic value of biomarkers using transcriptomic data in non-small-cell lung cancer. PLoS One. 2013;8, e82241.
Acknowledgments
This work was worked supported by the National Natural Science Foundation of China (No. 81172565 and No. 81202135).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding authors
Additional information
Synopsis for table of contents
In this study, we demonstrated that decreased BAP1 expression was associated with a shorter overall survival in gastric adenocarcinoma and may act an effective biomarker.
Shumei Yan and Fan He contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure 1
OS and FP curves for 593 gastric adenocarcinoma patients according to their BAP1 expression status (output from online Kaplan Meier plotter database). A. OS curves: patients with low (n=296) and high (n=297) expression levels of BAP1 respectively. B. FP curves: the first time when tumor progression was detected among patients with low and high expression levels of BAP1 respectively (JPEG 23.9 MB)
Rights and permissions
About this article
Cite this article
Yan, S., He, F., Luo, R. et al. Decreased expression of BRCA1-associated protein 1 predicts unfavorable survival in gastric adenocarcinoma. Tumor Biol. 37, 6125–6133 (2016). https://doi.org/10.1007/s13277-015-3983-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3983-0