Abstract
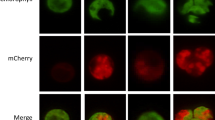
Recent developments in the genetic engineering of biotechnologically important microalgae—among them the diatom Phaeodactylum tricornutum—promise rapid progress in strain improvements for producing various valuable commodities. Nitrogen (N) depletion and starvation are commonly employed strategies to induce or enhance the formation of secondary carotenoids and storage lipids in microalgal biotechnology. The manipulation of gene expression for improving or rerouting the metabolic pathways requires the development of suitable stage-specific promoters to drive gene expression under desired conditions. Here, we describe the isolation, cloning, and characterization of novel P. tricornutum promoters that are either constitutively active in the presence or absence of nitrate as the sole N source, or upregulated under N starvation. An enhanced green fluorescent protein (eGFP), fused to a plasma membrane-anchoring sequence at the C terminus, was expressed under the control of several endogenous promoters, which were chosen based on previously reported gene expression levels under nitrate-limited or deprived conditions. Among the four promoters tested in this work, the ammonium transporter gene (AMT, PHATRDRAFT_27877) promoter strongly induced eGFP accumulation under N starvation, and the purine permease gene (PUP, PHATRDRAFT_52619) promoter strongly and constitutively drove eGFP accumulation, regardless of nitrate availability. The Act2 (actin-like 2, PHATRDRAFT_29136) and previously characterized DGAT1 gene promoters had relatively weak constitutive and late-inducible expression, respectively, under N starvation. The fucoxanthin-chlorophyll a/c-binding protein gene (fcpA) promoter used as a control was downregulated under N starvation. A comparison of quantitative real-time PCR gene expression data with western blot results suggested that controlled transcriptional regulation is important for efficient transgenic protein accumulation under N starvation. The results are of interest for the expression of transgenes in P. tricornutum for engineering oil production capacity and accumulating recombinant proteins.



Similar content being viewed by others
References
Abida H, Dolch L-J, Meï C, Villanova V, Conte M, Block MA, Finazzi G, Bastien O, Tirichine L, Bowler C, Rébeillé F, Petroutsos D, Jouhet J, Maréchal E (2015) Membrane glycerolipid remodeling triggered by nitrogen and phosphorus starvation in Phaeodactylum tricornutum. Plant Physiol 167:118–136
Alipanah L, Rohloff J, Winge P, Bones AM, Brembu T (2015) Whole-cell response to nitrogen deprivation in the diatom Phaeodactylum tricornutum. J Exp Bot 66:6281–6296
Alonso DL, Belarbi EH, Fernández-Sevilla JM, Rodríguez-Ruiz J, Molina Grima E (2000) Acyl lipid composition variation related to culture age and nitrogen concentration in continuous culture of the microalga Phaeodactylum tricornutum. Phytochemistry 54:461–471
Apt KE, Bhaya D, Grossman AR (1994) Characterization of genes encoding the light-harvesting proteins in diatoms: biogenesis of the fucoxanthin chlorophyll a/c protein complex. J Appl Phycol 6:225–230
Apt KE, Grossman AR, Kroth-Pancic PG (1996) Stable nuclear transformation of the diatom Phaeodactylum tricornutum. Mol Gen Genet 252:572–579
Benvenuti G, Bosma R, Klok AJ, Ji F, Lamers PP, Barbosa MJ, Wijffels RH (2015) Microalgal triacylglycerides production in outdoor batch-operated tubular PBRs. Biotechnol Biofuels 8:100. https://doi.org/10.1186/s13068-015-0283-2
Borowitzka MA (2013) High-value products from microalgae—their development and commercialisation. J Appl Phycol 25:743–756
Boussiba S, Khozin-Goldberg I, Shemesh Z, Leu S, Zarka A (2016) Algal promoters. PCT application Patent WO2016207893 A1
Bowler C, Allen AE, Badger JH et al (2008) The Phaeodactylum genome reveals the evolutionary history of diatom genomes. Nature 456:239–244
Cagnon C, Mirabella B, Nguyen HM, Beyly-Adriano A, Bouvet S, Cuiné S, Beisson F, Peltier G, Li-Beisson Y (2013) Development of a forward genetic screen to isolate oil mutants in the green microalga Chlamydomonas reinhardtii. Biotechnol Biofuels 6(1):178
Chu L, Ewe D, Río Bártulos C, Kroth PG, Gruber A (2016) Rapid induction of GFP expression by the nitrate reductase promoter in the diatom Phaeodactylum tricornutum. Peer J 4:e2344
Daboussi F, Leduc S, Maréchal A, Dubois G, Guyot V, Perez-Michaut C, Amato A, Falciatore A, Juillerat A, Beurdeley M, Voytas DF, Cavarec L, Duchateau P (2014) Genome engineering empowers the diatom Phaeodactylum tricornutum for biotechnology. Nat Commun 5:3831. https://doi.org/10.1038/ncomms4831
Dambek M, Eilers U, Breitenbach J, Steiger S, Büchel C, Sandmann G (2012) Biosynthesis of fucoxanthin and diadinoxanthin and function of initial pathway genes in Phaeodactylum tricornutum. J Exp Bot 63:5607–5612
De Martino A, Meichenin A, Shi J, Pan K, Bowler C (2007) Genetic and phenotypic characterization of Phaeodactylum tricornutum (Bacillariophyceae) accessions. J Phycol 43:992–1009
De Riso V, Raniello R, Maumus F, Rogato A, Bowler C, Falciatore A (2009) Gene silencing in the marine diatom Phaeodactylum tricornutum. Nucleic Acids Res 37(14):e96. https://doi.org/10.1093/nar/gkp448
Diner RE, Bielinski VA, Dupont CL, Allen AE, Weyman PD (2016) Refinement of the diatom episome maintenance sequence and improvement of conjugation-based DNA delivery methods. Front Bioeng Biotechnol 4:65. https://doi.org/10.3389/fbioe.2016.00065
Doyle JJ (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Falciatore A, Casotti R, Leblanc C, Abrescia C, Bowler C (1999) Transformation of nonselectable reporter genes in marine diatoms. Mar Biotechnol 1:239–251
Gahl RF, Tekle E, Zhu GA, Taraska JW, Tjandra N (2015) Acquiring snapshots of the orientation of trans-membrane protein domains using a hybrid FRET pair. FEBS Lett 589:885–889
Gardner R, Peters P, Peyton B, Cooksey KE (2011) Medium pH and nitrate concentration effects on accumulation of triacylglycerol in two members of the chlorophyta. J Appl Phycol 23:1005–1016
Goold H, Beisson F, Peltier G, Li-Beisson Y (2015) Microalgal lipid droplets: composition, diversity, biogenesis and functions. Plant Cell Rep 34:545–555
Griffiths MJ, Harrison STL (2009) Lipid productivity as a key characteristic for choosing algal species for biodiesel production. J Appl Phycol 21:493–507
Guihéneuf F, Leu S, Zarka A, Khozin-Goldberg I, Khalilov I, Boussiba S (2011) Cloning and molecular characterization of a novel acyl-CoA:diacylglycerol acyltransferase 1-like gene (PtDGAT1) from the diatom Phaeodactylum tricornutum. FEBS J 278:3651–3666
Hamilton ML, Haslam RP, Napier JA, Sayanova O (2014) Metabolic engineering of Phaeodactylum tricornutum for the enhanced accumulation of omega-3 long chain polyunsaturated fatty acids. Metab Eng. https://doi.org/10.1016/j.ymben.2013.12.003
Hempel F, Maier UG (2012) An engineered diatom acting like a plasma cell secreting human IgG antibodies with high efficiency. Microb Cell Factories 11:126
Hempel F, Lau J, Klingl A, Maier UG (2011) Algae as protein factories: expression of a human antibody and the respective antigen in the diatom. PLoS One 6:e28424
Hildebrand M (2005) Cloning and functional characterization of ammonium transporters from the marine diatom Cylindrotheca fusiformis (Bacillariophyceae). J Phycol 41:105–113
Hildebrand M, Dahlin K (2000) Nitrate transporter genes from the diatom Cylindrotheca fusiformis (Bacillariophyceae): mRNA levels controlled by nitrogen source and by the cell cycle. J Phycol 36:702–713
Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M, Darzins A (2008) Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J 54:621–639
Kadono T, Miyagawa-Yamaguchi A, Kira N, Tomaru Y, Okami T, Yoshimatsu T, Hou L, Ohama T, Fukunaga K, Okauchi M, Yamaguchi H, Ohnishi K, Falciatore A, Adachi M (2015) Characterization of marine diatom-infecting virus promoters in the model diatom Phaeodactylum tricornutum. Sci Rep 5:18708
Karas BJ, Diner RE, Lefebvre SC et al (2015) Designer diatom episomes delivered by bacterial conjugation. Nat Commun 6:6925
Kaye Y, Grundman O, Leu S, Zarka A, Zorin B, Didi-Cohen S, Khozin-Goldberg I, Boussiba S (2015) Metabolic engineering toward enhanced LC-PUFA biosynthesis in Nannochloropsis oceanica: overexpression of endogenous Δ12 desaturase driven by stress-inducible promoter leads to enhanced deposition of polyunsaturated fatty acids in TAG. Algal Res 11:387–398
Khozin-Goldberg I, Iskandarov U, Cohen Z (2011) LC-PUFA from photosynthetic microalgae: occurrence, biosynthesis, and prospects in biotechnology. Appl Microbiol Biotechnol 91:905–915
Leu S, Boussiba S (2014) Advances in the production of high-value products by microalgae. Ind Biotechnol 10:169–183
Levitan O, Dinamarca J, Zelzion E, Lun DS, Guerra LT, Kim MK, Kim J, Van Mooy BA, Bhattacharya D, Falkowski PG (2015) Remodeling of intermediate metabolism in the diatom Phaeodactylum tricornutum under nitrogen stress. Proc Natl Acad Sci 112:412–417
Levraud J-P, Boudinot P, Colin I, Benmansour A, Peyrieras N, Herbomel P, Lutfalla G (2007) Identification of the zebrafish IFN receptor: implications for the origin of the vertebrate IFN system. J Immunol 178:4385–4394
Liu X, Hempel F, Stork S, Bolte K, Moog D, Heimerl T, Maier UG, Zauner S (2016) Addressing various compartments of the diatom model organism Phaeodactylum tricornutum via sub-cellular marker proteins. Algal Res 20:249–257
Maheswari U, Montsant A, Goll J, Krishnasamy S, Rajyashri KR, Patell VM, Bowler C (2005) The diatom EST database. Nucleic Acids Res 33:D344–D347
Maheswari U, Jabbari K, Petit J-L et al (2010) Digital expression profiling of novel diatom transcripts provides insight into their biological functions. Genome Biol 11:R85
Matthijs M, Fabris M, Broos S, Vyverman W, Goossens A (2015) Profiling of the early nitrogen stress response in the diatom Phaeodactylum tricornutum reveals a novel family of RING-domain transcription factors. Plant Physiol 170:489–498
Niu YF, Yang ZK, Zhang MH, Zhu CC, Yang WD, Liu JS, Li HY (2012) Transformation of diatom Phaeodactylum tricornutum by electroporation and establishment of inducible selection marker. BioTechniques 52(6). https://doi.org/10.2144/000113881
Nymark M, Sharma AK, Sparstad T, Bones AM, Winge P (2016) A CRISPR/Cas9 system adapted for gene editing in marine algae. Sci Rep 6:24951
Pal D, Khozin-Goldberg I, Cohen Z, Boussiba S (2011) The effect of light, salinity, and nitrogen availability on lipid production by Nannochloropsis sp. Appl Microbiol Biotechnol 90:1429–1441
Peled E, Leu S, Zarka A, Weiss M, Pick U, Khozin-Goldberg I, Boussiba S (2011) Isolation of a novel oil globule protein from the green alga Haematococcus pluvialis (Chlorophyceae). Lipids 46:851–861
Popko J, Herrfurth C, Feussner K, Ischebeck T, Iven T, Haslam R, Hamilton M, Sayanova O, Napier J, Khozin-Goldberg I (2016) Metabolome analysis reveals betaine lipids as major source for triglyceride formation, and the accumulation of sedoheptulose during nitrogen-starvation of Phaeodactylum tricornutum. PLoS One 11(10):e0164673
Poulsen N, Kröger N (2005) A new molecular tool for transgenic diatoms: control of mRNA and protein biosynthesis by an inducible promoter-terminator cassette. FEBS J 272:3413–3423
Pulz O, Gross W (2004) Valuable products from biotechnology of microalgae. Appl Microbiol Biotechnol 65:635–648
Rodolfi L, Chini Zittelli G, Bassi N, Padovani G, Biondi N, Bonini G, Tredici M (2009) Microalgae for oil: strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol Bioeng 102:100–112
Rosenwasser S, Graff van Creveld S, Schatz D, Malitsky S, Tzfadia O, Aharoni A, Levin Y, Gabashvili A, Feldmesser E, Vardi A (2014) Mapping the diatom redox-sensitive proteome provides insight into response to nitrogen stress in the marine environment. Proc Natl Acad Sci U S A 111:2740–2745
Sakaue K, Harada H, Matsuda Y (2008) Development of gene expression system in a marine diatom using viral promoters of a wide variety of origin. Physiol Plant 133:59–67
Sapriel G, Quinet M, Heijde M, Jourdren L, Tanty V, Luo G, Le Crom S, Lopez PJ (2009) Genome-wide transcriptome analyses of silicon metabolism in Phaeodactylum tricornutum reveal the multilevel regulation of silicic acid transporters. PLoS One 4:e7458
Seo S, Jeon H, Hwang S, Jin E, Chang KS (2015) Development of a new constitutive expression system for the transformation of the diatom Phaeodactylum tricornutum. Algal Res 11:50–54
Shemesh Z, Leu S, Khozin-Goldberg I, Didi-Cohen S, Zarka A, Boussiba S (2016) Inducible expression of Haematococcus oil globule protein in the diatom Phaeodactylum tricornutum: association with lipid droplets and enhancement of TAG accumulation under nitrogen starvation. Algal Res 18:321–331
Siaut M, Heijde M, Mangogna M, Montsant A, Coesel S, Allen A, Manfredonia A, Falciatore A, Bowler C (2007) Molecular toolbox for studying diatom biology in Phaeodactylum tricornutum. Gene 406:23–35
Valenzuela J, Mazurie A, Carlson RP, Gerlach R, Cooksey KE, Peyton BM, Fields MW (2012) Potential role of multiple carbon fixation pathways during lipid accumulation in Phaeodactylum tricornutum. Biotechnol Biofuels 5(1):40
Valenzuela J, Carlson RP, Gerlach R, Cooksey K, Peyton BM, Bothner B, Fields MW (2013) Nutrient resupplementation arrests bio-oil accumulation in Phaeodactylum tricornutum. Appl Microbiol Biotechnol 97:7049–7059
Vanier G, Hempel F, Chan P, Rodamer M, Vaudry D, Maier UG, Lerouge P, Bardor M (2015) Biochemical characterization of human anti-hepatitis B monoclonal antibody produced in the microalgae Phaeodactylum tricornutum. PLoS One 10(1):e0139282
Weyman PD, Beeri K, Lefebvre S, Rivera J, McCarthy JK, Heuberger AL, Peers G, Allen AE, Dupont CL (2014) Inactivation of Phaeodactylum tricornutum urease gene using TALEN-based targeted mutagenesis. Plant Biotechnol J 13:460–470
Wijffels RH, Barbosa MJ (2010) An outlook on microalgal biofuels. Science 329:796–799
Yoshinaga R, Niwa-Kubota M, Matsui H, Matsuda Y (2014) Characterization of iron-responsive promoters in the marine diatom Phaeodactylum tricornutum. Mar Genomics 16:55–62
Zaslavskaia LA, Lippmeier JC, Kroth PG, Grossman AR, Apt KE (2000) Transformation of the diatom Phaeodactylum tricornutum with a variety of selectable marker and reporter genes. J Phycol 386:379–386
Zaslavskaia LA, Lippmeier JC, Shih C, Ehrhardt D, Grossman AR, Apt KE (2001) Trophic conversion of an obligate photoautotrophic organism through metabolic engineering. Science 292:2073–2075
Zendejas FJ, Benke PI, Lane PD, Simmons BA, Lane TW (2012) Characterization of the acylglycerols and resulting biodiesel derived from vegetable oil and microalgae (Thalassiosira pseudonana and Phaeodactylum tricornutum). Biotechnol Bioeng 109:1146–1154
Zhang FL, Casey PJ (1996) Protein prenylation: molecular mechanisms and functional consequences. Annu Rev Biochem 65:241–269
Zhang L, Gurskaya NG, Merzlyak EM, Staroverov DB, Mudrik NN, Samarkina ON, Vinokurov LM, Lukyanov S, Lukyanov KA (2007) Method for real-time monitoring of protein degradation at the single cell level. BioTechniques 42:446–450
Zienkiewicz K, Z-Y D, Ma W, Vollheyde K, Benning C (2016) Stress-induced neutral lipid biosynthesis in microalgae—molecular, cellular and physiological insights. Biochim Biophys Acta Mol Cell Biol Lipids 9:1269–1281
Acknowledgements
Z.A.A. acknowledges receipt of a PhD fellowship and a Short-Term Post-Doctoral Fellowship from the Kreitman School of Advanced Graduate Studies at Ben-Gurion University of the Negev.
Funding
This work was financially supported by the European Commission’s Seventh Framework Programme for Research and Technology Development (EC-FP7), project Fuel4ME (Grant No. 308983) and GIAVAP (Grant No. 266401), and by the I-CORE Solar Fuels Program of the Planning and Budgeting Committee and The Israel Science Foundation (Grant no. 152/11).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(DOCX 139 kb)
Rights and permissions
About this article
Cite this article
Adler-Agnon (Shemesh), Z., Leu, S., Zarka, A. et al. Novel promoters for constitutive and inducible expression of transgenes in the diatom Phaeodactylum tricornutum under varied nitrate availability. J Appl Phycol 30, 2763–2772 (2018). https://doi.org/10.1007/s10811-017-1335-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-017-1335-8