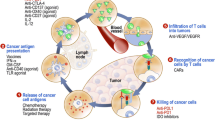
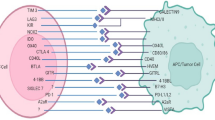
Abstract
Background
Photodynamic therapy (PDT) plays an immunoregulatory role in tumours. Here, we conducted a retrospective patient analysis to evaluate the effectiveness of PDT plus immune checkpoint inhibitors (ICIs) in gastric cancer. Further, we performed a dynamic analysis of gastric cancer patients receiving PDT to clarify its effects on anti-tumour immunity.
Methods
Forty ICI-treated patients that received PDT or not were retrospectively analysed. Five patients with gastric adenocarcinoma were enrolled for sample collection before and after PDT. Single-cell RNA/T cell receptor (TCR) sequencing, flow cytometry and histological exanimation were used to analyse the collected specimens.
Results
Patients in PDT group had a significantly better OS after ICI treatment than those in No PDT group. Single-cell analysis identified ten cell types in gastric cancer tissues and four sub-populations of T cells. Immune cell infiltration increased in the tumours after PDT and the circular immune cells showed consistent alterations. TCR analysis revealed a specific clonal expansion after PDT in cytotoxic T lymphocytes (CTL), but a constriction in Tregs. The B2M gene is upregulated in tumour cells after PDT and is associated with immune cell infiltration. Several pathways involving the positive regulation of immunity were enriched in tumour cells in the post-PDT group. The interactions following PDT were increased between tumour cells and effector cells but decreased between Tregs and other immune cells. Some co-stimulatory signaling emerged, whereas co-inhibitory signaling disappeared in intercellular communication after PDT.
Conclusions
PDT elicits an anti-tumour response through various mechanisms and is promising as an adjuvant to enhance ICI benefit.







Similar content being viewed by others
Availability of data and materials
All data supporting this study have been presented in the full text or online supplementary files. Other relevant materials can be obtained on reasonable request to the corresponding author.
Abbreviations
- ICIs:
-
Immune checkpoint inhibitors
- PDT:
-
Photodynamic therapy
- ICD:
-
Immunogenic cell death
- scRNA/TCR-seq:
-
Single-cell RNA/T cell receptor (TCR) sequencing
- OS:
-
Overall survival
- HE:
-
Hematoxylin–eosin
- PBS:
-
Phosphate buffered saline
- UMIs:
-
Unique molecule identifiers
- PCA:
-
Principal component analysis
- t-SNE:
-
T-distributed stochastic neighbour embedding
- GO:
-
Gene ontology
- KEGG:
-
Kyoto encyclopedia of genes and genomes
- TCGA:
-
The cancer genome atlas
- NKT:
-
Natural killer T cells
- Th:
-
T helper cells
- Treg:
-
CD4+ regulatory T cells
- CTL:
-
CD8+ cytotoxic T cells
- HRs:
-
Hazard ratios
- CIs:
-
Confidence intervals
- HSC:
-
Haematopoietic stem cells
- TSAs:
-
Tumour specific antigens
References
Yang L, Ying X, Liu S, Lyu G, Xu Z, Zhang X, et al. Gastric cancer: epidemiology, risk factors and prevention strategies. Chin J Cancer Res = Chung-kuo yen cheng yen chiu. 2020;32(6):695–704.
Kahraman S, Yalcin S. Recent advances in systemic treatments for HER-2 positive advanced gastric cancer. Onco Targets Ther. 2021;14:4149–62.
Committee CGW. The Chinese Society of clinical oncology (CSCO): clinical guidelines for the diagnosis and treatment of gastric cancer, 2022. Beijing: People’s Medical Publishing House; 2022.
Chyuan IT, Chu CL, Hsu PN. Targeting the tumor microenvironment for improving therapeutic effectiveness in cancer immunotherapy: focusing on immune checkpoint inhibitors and combination therapies. Cancers. 2021;13(6):1188.
Nath S, Obaid G, Hasan T. The course of immune stimulation by photodynamic therapy: bridging fundamentals of photochemically induced immunogenic cell death to the enrichment of T-cell repertoire. Photochem Photobiol. 2019;95(6):1288–305.
Falk-Mahapatra R, Gollnick SO. Photodynamic therapy and immunity: an update. Photochem Photobiol. 2020;96(3):550–9.
Beltrán Hernández I, Yu Y, Ossendorp F, Korbelik M, Oliveira S. Preclinical and clinical evidence of immune responses triggered in oncologic photodynamic therapy: clinical recommendations. J Clin Med. 2020;9(2):333.
Cavin S, Gkasti A, Faget J, Hao Y, Letovanec I, Reichenbach M, et al. Low-dose photodynamic therapy promotes a cytotoxic immunological response in a murine model of pleural mesothelioma. Eur J Cardio-thorac Surg Off J Eur Assoc Cardio-thorac Surg. 2020;58(4):783–91.
Kleinovink JW, Fransen MF, Löwik CW, Ossendorp F. Photodynamic-immune checkpoint therapy eradicates local and distant tumors by CD8(+) T cells. Cancer Immunol Res. 2017;5(10):832–8.
Zhang M, Hu S, Min M, Ni Y, Lu Z, Sun X, et al. Dissecting transcriptional heterogeneity in primary gastric adenocarcinoma by single cell RNA sequencing. Gut. 2021;70(3):464–75.
Kumar V, Ramnarayanan K, Sundar R, Padmanabhan N, Srivastava S, Koiwa M, et al. Single-cell atlas of lineage states, tumor microenvironment, and subtype-specific expression programs in gastric cancer. Cancer Discov. 2022;12(3):670–91.
Bassez A, Vos H, Van Dyck L, Floris G, Arijs I, Desmedt C, et al. A single-cell map of intratumoral changes during anti-PD1 treatment of patients with breast cancer. Nat Med. 2021;27(5):820–32.
Li T, Fan J, Wang B, Traugh N, Chen Q, Liu JS, et al. TIMER: a web server for comprehensive analysis of tumor-infiltrating immune cells. Can Res. 2017;77(21):e108–10.
Efremova M, Vento-Tormo M, Teichmann SA, Vento-Tormo R. Cell PhoneDB: inferring cell-cell communication from combined expression of multi-subunit ligand-receptor complexes. Nat Protoc. 2020;15(4):1484–506.
Li F, Li C, Cai X, Xie Z, Zhou L, Cheng B, et al. The association between CD8+ tumor-infiltrating lymphocytes and the clinical outcome of cancer immunotherapy: a systematic review and meta-analysis. EClinicalMedicine. 2021;41:101134.
Gide TN, Quek C, Menzies AM, Tasker AT, Shang P, Holst J, et al. Distinct immune cell populations define response to anti-PD-1 monotherapy and anti-PD-1/anti-CTLA-4 combined therapy. Cancer Cell. 2019;35(2):238-55.e6.
Kim KH, Cho J, Ku BM, Koh J, Sun JM, Lee SH, et al. The first-week proliferative response of peripheral blood PD-1(+)CD8(+) T cells predicts the response to anti-PD-1 therapy in solid tumors. Clin Cancer Res Off J Am Assoc Cancer Res. 2019;25(7):2144–54.
Kamphorst AO, Pillai RN, Yang S, Nasti TH, Akondy RS, Wieland A, et al. Proliferation of PD-1+ CD8 T cells in peripheral blood after PD-1-targeted therapy in lung cancer patients. Proc Natl Acad Sci USA. 2017;114(19):4993–8.
Vafaei S, Zekiy AO, Khanamir RA, Zaman BA, Ghayourvahdat A, Azimizonuzi H, et al. Combination therapy with immune checkpoint inhibitors (ICIs); a new frontier. Cancer Cell Int. 2022;22(1):2.
Yan YD, Cui JJ, Fu J, Su YJ, Chen XY, Gu ZC, et al. A Network comparison on safety profiling of immune checkpoint inhibitors in advanced lung cancer. Front Immunol. 2021;12:760737.
Theodoraki MN, Lorenz K, Lotfi R, Fürst D, Tsamadou C, Jaekle S, et al. Influence of photodynamic therapy on peripheral immune cell populations and cytokine concentrations in head and neck cancer. Photodiagn Photodyn Ther. 2017;19:194–201.
Hass R, Jacobs R, Kaufmann AM, Hillemanns P, Soergel P. Sensitization of immune cells following hexylaminolevulinate photodynamic therapy of cervical intraepithelial neoplasia. Photodiagn Photodyn Ther. 2017;17:82–6.
Yu Y, Yu R, Wang N, Bai Y, Shi Q, Maswikiti EP, et al. Photodynamic therapy in combination with immune checkpoint inhibitors plus chemotherapy for first-line treatment in advanced or metastatic gastric or gastroesophageal junction cancer: a phase 2–3 clinical trial protocol. Front Pharmacol. 2023;14:1063775.
Rhee JK, Jung YC, Kim KR, Yoo J, Kim J, Lee YJ, et al. Impact of tumor purity on immune gene expression and clustering analyses across multiple cancer types. Cancer Immunol Res. 2018;6(1):87–97.
Lou S, Zhang J, Yin X, Zhang Y, Fang T, Wang Y, et al. Comprehensive characterization of tumor purity and its clinical implications in gastric cancer. Front Cell Dev Biol. 2021;9:782529.
Jhunjhunwala S, Hammer C, Delamarre L. Antigen presentation in cancer: insights into tumour immunogenicity and immune evasion. Nat Rev Cancer. 2021;21(5):298–312.
Wang H, Liu B, Wei J. Beta2-microglobulin(B2M) in cancer immunotherapies: biological function, resistance and remedy. Cancer Lett. 2021;517:96–104.
Chen DS, Mellman I. Elements of cancer immunity and the cancer-immune set point. Nature. 2017;541(7637):321–30.
Acknowledgements
The authors would like to thank BMKCloud (http://www.biocloud.net/) for providing supports in data process, management and figure drawing. We thank Chen Yu, Tianliang Lu, and Rui Zhang for their advice in bioinformatics analysis and Xiaohong Liu for her assistance in sample collection. We also thank the patients and their families who donated the tissue and blood samples in this study.
Funding
This work was supported by the Key Project of Science and Technology in Gansu province (22ZD6FA054 and 19ZD2WA001), Key Talents Project of Gansu Province (2019RCXM020), Science and Technology Project of Chengguan District of Lanzhou City (2020SHFZ0039 and 2020JSCX0073), and Medical Innovation and Development Project of Lanzhou University (lzuyxcx-2022-160, lzuyxcx-2022-45, and lzuyxcx-2022-88).
Author information
Authors and Affiliations
Contributions
YY and HC: had the idea of this study. YY and HC: conceived and designed the study. YY: performed most of the experiments, including single-cell sequencing, flow cytometry, and data analysis. BX, LX, NW, RY, and LG: contributed to the recruitment and management of patients. TLD, EPM, BX, and YPW: performed sample collection. HYL and BHG: helped with the processing and preparation of single-cell suspensions. CHM, BFW, and XYW: collected patient information and made the follow-up. YY and TZ: generated the statistical strategy. YY: visualised and interpreted the results and drafted the manuscript. YPB, LX, PZ and HC: provided clinical perspectives for the interpretation of results. HC and LX: reviewed and revised the manuscript. HC obtained funding to support all aspects of this study. YY and HC: have read and verified the underlying data. All authors read and approved the final version of the manuscript for submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
This study was approved by the Institutional Research Ethics Committee of the Lanzhou University Second Hospital (2021A-146). Informed consent was obtained from all included patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, Y., Xu, B., Xiang, L. et al. Photodynamic therapy improves the outcome of immune checkpoint inhibitors via remodelling anti-tumour immunity in patients with gastric cancer. Gastric Cancer 26, 798–813 (2023). https://doi.org/10.1007/s10120-023-01409-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10120-023-01409-x