Summary
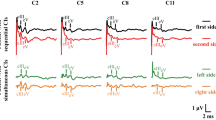
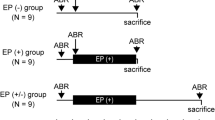
Reversible conductive hearing loss created during the first 4 weeks post partum caused marked alterations in the maturation of binaural interaction components in the auditory brainstem responses of guinea pigs. In untreated control animals all three components investigated demonstrated postnatal development in terms of latency shortening that was completed during the first 3 weeks of life. Plugging of both external ear canals caused a significant delay in the maturation of the late component DN2, where latency values of the controls were reached only 2 weeks after the end of the treatment, i.e. after 6 weeks of life. Monaural deprivation likewise led to a retarded development of peak latencies during the phase of imbalanced sensory imput. After the end of the one-sided conductive hearing loss the maturation process was markedly enhanced, even resulting in latency values for DN2 and DP1 that were significantly shorter than those of the controls. This phenomenon persisted until the end of the study period and was the case for both plugged and untreated ears in this group of animals. The time course of latencies in two other groups of experimental animals which were deprived in the same way as adults suggests that the effects observed are due to a sensitive period in the maturation process of the auditory pathway.
Similar content being viewed by others
References
Clements M, Kelly JB (1978) Auditory spatial response of young guinea pigs (Cavia porcellus) during and after ear blocking. J Comp Physiol 92:34–44
Clopton BM, Silverman MS (1977) Plasticity of binaural interaction. II. Critical period and changes in midline response. J Neurophysiol 40:1275–1280
Dum N (1984) Postnatal development of the auditory evoked brainstem potentials in the guinea pig. Acta Otolaryngol (Stockh) 97:63–68
Feng AS, Rogowski BA (1980) Effects of monaural and binaural occlusion on the morphology of neurons in the medial superior olivary nucleus of the rat. Brain Res 189:530–534
Guerit JM (1985) Application of surface-recorded auditory evoked potentials for the early diagnosis of hearing loss in neonates and premature infants. Acta Otolaryngol 421 [Suppl] (Stockh):68–76
Henry KR (1983) Abnormal auditory development resulting from exposure to ototoxic chemicals, noise, and auditory restriction. In: Romand R (ed) Development of auditory and vestibular systems. Academic Press, New York, pp 273:308
Hosford-Dunn H, Mendelson T, Salamy A (1981) Binaural interactions in short-latency evoked potentials of neonates. Audiology 20:394–408
Jen PHS, Sun X (1990) Influence of monaural plugging on postnatal development of auditory spatial sensitivity of inferior colliculus neurons of the big brown bat, Eptesicus fuscus. Chin J Physiol 33:231–246
Killackey HP, Ryugo DK (1977) Effects of neonatal auditory system damage on the structure of the inferior colliculus of the rat. Anat Rec 187:624
Knudsen EI, Esterly SD, Knudsen PF (1984) Monaural occlusion alters sound localization during a sensitive period in the barn owl. J Neurosci 4:1001–1011
Knudsen EI, Knudsen PF, Esterly SD (1984) A critical period of the recovery of sound localization accuracy following monaural occlusion in the barn owl. J Neurosci 4:1012–1020
Moore DR (1983) Development of inferior colliculus and binaural audition. In: Romand R (ed) Development of auditory and vestibular systems. Academic Press, New York, pp 121–166
Moore DR, Irvine DRF (1981) Plasticity of binaural interaction in the cat inferior colliculus. Brain Res 208: 198–202
Nordeen KW, Killackey HP, Kitzes LM (1983) Ascending projections in the inferior colliculus following unilateral cochlear ablation in the neonatal gerbil, Meriones unguiculatus. J Comp Neurol 214:144–153
Rotteveel JJ, Colon EJ, Notermans SLH, Stoelinga GBA, Visco YM (1985) The central auditory conduction at term and three months after birth. Scand Audiol 14:179–186
Silverman MS, Clopton BM (1977) Plasticity of binaural interaction. I. Effect of early auditory deprivation. J Neurophysiol 40:1266–1280
Trune DR (1982) Influence of neonatal cochlear removal on the development of mouse cochlear nucleus. I. Number, size, and density of its neurons. J Comp Neurol 209:409–424
Uziel A (1985) Non-genetic factors affecting hearing development. Acta Otolaryngol 421 [Suppl] (Stock): 57–61
Walger M, Ferreira P, Laska M, Schneider I, Wedel H von (1989) Einfluß binauraler Schalleitungsstörung auf die Reifung akustisch evozierter Potentiale (HSP, MLR) beim Meerschweinchen. Laryngol Rhinol Otol 68:626–631
Wedel H von, Schauseil-Zipf U, Daring WH (1988) Hörscreening bei Neugeborenen und Säuglingen mittels Ableitung akustisch evozierter Hirnstammpotentiale. Laryngol Rhinol Otol 67:307–311
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Laska, M., Walger, M., Schneider, I. et al. Maturation of binaural interaction components in auditory brainstem responses of young guinea pigs with monaural or binaural conductive hearing loss. Eur Arch Otorhinolaryngol 249, 325–328 (1992). https://doi.org/10.1007/BF00179382
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00179382