Abstract
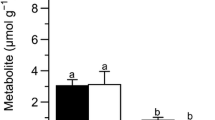
The metabolic effects of evaporative water loss at 5 °C were assessed for both fall- and spring-collected spring peepersPsuedacris crucifer. Frogs readily endured the loss of 50% of total body water. During dehydration organ water content was defended with no change in water content in skeletal muscle, gut, and kidney of 50% dehydrated frogs and reduced water content in liver, brain and heart. Dehydration stimulated a rapid and massive increase in liver glucose production. In fall-collected frogs liver glucose rose by 120-fold to 2690±400 nmol · mg protein-1 or 220 μmol · g ww-1 in 50% dehydrated frogs and glucose in other organs increased by 2.6- to 60-fold. Spring-collected frogs showed the same qualitative response to dehydration although absolute glucose levels were lower, rising maximally by 8.4-fold in liver. Glucose synthesis was supported by glycogenolysis in liver and changes in the levels of glycolytic intermediates in liver indicated that an inhibitory block at the phosphofructokinase locus during desiccation helped to divert hexose phosphates into the production of glucose. Liver energy status (ATP, total adenylates, energy charge) was maintained even after the loss of 35% of total body water but at 50% dehydration all parameters showed a sharp decline; for example, energy charge fell from about 0.85 to 0.42. Severe dehydration also led to an accumulation of lactate in four organs, probably hypoxia-induced the to impaired circulation. The hyperglycemic response ofP. crucifer to dehydration mimics the cryoprotectant synthesis response seen during freezing of this freeze-tolerant frog, suggesting that these share a common regultory mechanism and that the cryoprotectant response may have arisen out of pre-existing volume regulatory responses of amphibians. The hyperglycemic response to dehydration might also be utilized during winter hibernation to help retard body water loss by raising the osmolality of the body fluids in situations where hibernaculum conditions become dry.
Similar content being viewed by others
Abbreviations
- bin:
-
body mass
- bw:
-
body water
- CrP:
-
creatine phosphate
- dw:
-
dry weight
- F6P:
-
fructose-6-phosphate
- FBP:
-
fructose-1,6-bisphosphate
- G6P:
-
glucose-6-phosphate
- PEP:
-
phosphoenolpyruvate
- PFK:
-
phosphofructokinase
- PYR:
-
pyruvate
- ww:
-
wet weight
References
Churchill TA (1992) Metabolic control of freeze tolerance invertebrates. Ph.D. thesis, Carleton University, Ottawa
Churchill TA, Storey KB (1993) Dehydration tolerance in wood frogs: a new perspective on the development of amphibian freeze tolerance. Am J Physiol 265:R1324-R1332
Churchill TA, Storey KB (1994) Metabolic effects of dehydration on an aquatic frog,Rana pipiens. J Exp Biol (in press)
Gatten R (1987) Activity metabolism of anuran amphibians: tolerance to dehydration. Physiol Zool 60:576–585
Hillman S (1978) The roles of oxygen delivery and electrolyte levels in the dehydrational death ofXenopus laevis. J Comp Physiol 128:169–175
Hillman S (1980) Physiological correlates of differential dehydration tolerance in anuran amphibians. Copeia 1980:125–129
Hillman S (1987) Dehydrational effects on cardiovascular and metabolic capacity in two amphibians. Physiol Zool 60:608–613
Hillman S (1988) Dehydrational effects on brain and cerebrospinal fluid electrolytes in two amphibians. Physiol Zool 61:254–259
Keppler D, Decker K (1974) Glycogen: determination with amyloglucosidase. In: Bergmever HU (ed) Methods of enzymatic analysis. Academic Press, New York, pp 1127–1131
Lowry OH, Passonneau JV (1972) A flexible system of enzymatic analysis. Academic Press, New York
Pinder AW, Storey KB, Ultsch GR (1992) Estivation and hibernation. In: Feder ME, Burggren WW (eds) Environmental physiology of the amphibians. University of Chicago Press, Chicago, pp 250–274
Shoemaker VH (1992) Exchange of water, ions and respiratory gases in terrestrial amphibians. In: Feder ME, Burggren WW (eds) Environmental physiology of the amphibians. University of Chicago Press, Chicago, pp 125–150
Shpun S, Hoffman J, Katz U (1992) Anuran amphibia which are not acclimable to high salt, tolerate high plasma urea. Comp Biochem Physiol 103A:473–477
Storey JM, Storey KB (1985) Triggering of cryoprotectant synthesis by the initiation of ice nucleation in the freeze tolerant frog,Rana sylvatica. J Comp Physiol B 156:191–195
Storey KB (1987) Glycolysis and the regulation of cryoprotectant synthesis in liver of the freeze tolerant wood frog. J Comp Physiol B 157:373–380
Storey KB, Storey JM (1984) Biochemical adaptation for freezing tolerance in the wood frog,Rana sylvatica. J Comp Physiol B 155:29–36
Storey KB, Storey JM (1986a) Freeze tolerance and intolerance as strategies of winter survival in terrestrially hibernating amphibians. Comp Biochem Physiol 83A:613–617
Storey KB, Storey JM (1986b) Freeze tolerant frogs: cryoprotectants and tissue metabolism during freeze-thaw cycles. Can J Zool 64:49–56
Storey KB, Storey JM (1987) Persistence of freeze tolerance in terrestrially hibernating frogs after spring emergence. Copeia 1987:720–726
Storey KB, Storey JM (1992) Natural freeze tolerance in ectothermic vertebrates. Annu Rev Physiol 54:619–637
Thorson TB, Svihla A (1943) Correlation of the habitats of amphibians with their ability to survive the loss of body water. Ecology 24:274–281
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Churchill, T.A., Storey, K.B. Effects of dehydration on organ metabolism in the frogPseudacris crucifer: hyperglycemic responses to dehydration mimic freezing-induced cryoprotectant production. J Comp Physiol B 164, 492–498 (1994). https://doi.org/10.1007/BF00714587
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00714587