Abstract
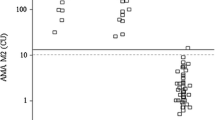
Aim of this study was to determine and furthercharacterize the serum aminopeptidase-M in children withliver diseases. Based on our new assay, we have showntwo fractions of the enzyme. Activity of the first fraction is expressed in undiluted serumat pH adjusted from 8.5 (pH of storaged serum) to 7.4.Activity of the second fraction (cryptic activity)appears in the serum (pH 7.4) as a result of dilution and/or addition of aniline naphthalene sulfonicacid. In children with Alagille syndrome, extrahepaticbiliary duct atresia, Byler's disease, and acutehepatitis due to hepatitis B virus infection, activities of both fractions are highly elevated ascompared to healthy children or those with chronic viralhepatitis. Moreover, serum aminopeptidase-M seems toreflect other aspects of the pathological process than those reflected by the alanine aminotransferaseand gamma-glutamyltranspeptidase. Due to increasedactivity and broad substrate specificity, the enzymeseems to be also a cofactor of cholestasis andhepatitis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
REFERENCES
Hersh LB, Aboukhair N, Watson S: Immunocytochemical localisation of aminopeptidase-M in rat brain and periphery: Relationship of the enzyme localisation and enkephalin metabolism. Peptides 8:523-532, 1987
Roques BP, Beaumont A, Dauge V, Fournie-Zaluski M-C: Peptidase inactivation of enkephalins: Design of inhibitors and biochemical, pharmacological and clinical applications. In Handbook of Experimental Pharmacology. A Hertz (ed). Berlin, Springer-Verlag, 1993, pp 547-584
Kenny AJ, Hooper NM: Peptidases involved in the metabolism of bioactive peptides. In Degradation of Bioactive Substances: Physiology and Pathophysiology. JH Henrickson (ed). Boca Raton, Florida, CRC Press, 1991, pp 47-79
Peters JE, Nilius R, Otto L: Elektrophoretische Varianten der Alanin–Aminopeptidase in Se rum be i hepatobi l iären Erkrankungen. Clin Chim Acta 45:177-187, 1973
Tokioka-Terao M, Hiwada K, Kokubu T: A radioimmunoassay for the measurement of aminopeptidase (microsomal) in human serum. Enzyme 33:181-187, 1985
Sanderink G-J, Artur Y, Paille F, Siest G: Clinical significance of a new isoform of serum alanine aminopeptidase; relationship with liver disease and alcohol consumption. Clin Chim Acta 179:23-32, 1989
Favaloro EJ, Browning T, Nandurkar H: The hepatobiliary disease marker serum alanine aminopeptidase predominantly comprises an isoform of the haematological myeloid differentiation antigen and leukaemia marker CD-13/gp150. Clin Chim Acta 220:81-90, 1993
Sidorowicz W, Hsia W, Masle j-Zownir O, Behal FJ: Multiple molecular forms of human alanine aminopeptidase: Immunological properties. Clin Chim Acta 107:245-256, 1980
Ashmun RA, Look AT: Metalloproteaze activity of CD13/ aminopeptidase-N on the surface of human myeloid cells. Blood 75:462-469, 1990
Look AT, Ashmun RA, Shapiro LH, Peiper SC: Human myeloid plasma membrane glycoprote in CD-13 (gp150) is identical to aminopeptidase-N. J Clin Invest 83:1299-1307, 1989
Favaloro EJ, Browning T, Facey D: CD13 (GP 150; aminopeptidase-N): predominant functional activity in blood cells is localised to plasma and is not cell-surface associated. Exp Hematol 21:1695-1701, 1993
Yeager CL, Ashmun RA, Williams RK, Cardellichio CB, Shapiro LH, Look AT, Holmes KV: Human aminopeptidase-N is a receptor for human coronavirus 229E. Nature 357:420-422, 1992
Sanderink G-J, Artur Y, Schiele F, Gueguen R, Siest G: Alanine aminopeptidase in serum: Biological variations and reference limits. Clin Chem 34 (7):1422-1426, 1988
Gibson AM, Biggins JA, Lauffart B, Mantele D, McDermott JR: Human brain leucyl aminopeptidase: Isolation, characterisation and specificity against some neuropeptides. Neuropeptides 19:163-168, 1991
Mizutani S, Goto K, Nomura S, Ino K, Kikkawa F, Kurauchi D, Goldstein D, Tomoda Y: Possible action of human placental aminopeptidase-N in feto-placental unit. Res Commun Chem Pathol Pharmacol 82:65-80, 1993
Kenny AJ, O'Hare MJ, Gusterson BA: Cell-surface peptidases as modulators of growth and differentiation. Lancet 30:785-787, 1989
Boarder MR, McArdle W: Breakdown of small enkephalin deriviates and adrenal peptide E by human plasma. Biochem Pharmacol 35:1043-1047, 1986
Lolu K, Lampelo S, Vanha-Perttula T: Characte risation of three aminopeptidases purified from maternal serum. Biochim Biophys Acta 873:190-197, 1986
Venturelli F, Roscetti G, Roda LG: Mechanism protecting plasma peptides from enzyme hydrolysis: A comparative study. Comp Biochem Physiol 87B:485-488, 1987
Martinez JL, Weinberger SB: Enkephalin hydrolysis in plasma is highly correlated with escape performance in the rat. Behav Neurosci 102:404-408, 1988
Weinberger SB, Martinez JL: Characterisation of hydrolysis of [Leu] enkephalin and D-ala2-[1-leu]enkephalin in rat plasma. J Pharmacol Exp Ther 102:404-408, 1988
Shibanoki S, Weinberger SB, Ishikawa K, Martinez JL: Sensitive method for measuring hydrolysis of enkephalins in plasma, using high-performance liquid chromatography with electrochemical detection. J Chromatogr 532:249-259, 1990
Berder IF, Hirsh EM, Tuchman AJ, Wards PE: N-terminal degradation of low molecular weigh opioid peptides in human cerebrospinal fluid. Biochem Pharmacol 40:465-472, 1990
Shibanoki S, Weinberger SB, Ishikawa K, Martinez JL: Further characterisation of the in vitro hydrolysis of [Leu]-and [Met]enkephalin in rat plasma: HPLC-ECD measurements of substrate and metabolite concentrations. Regul Pept 32:267-278, 1991
Jonak PH, Schulteis G, Martinez JL Jr: Uptake and me tabolism of [3H]-Leu-enkephalin following either its intraperitoneal or subcutaneous administration to mice. Peptides 13:551-555, 1992
Schulteis G, Martinez JL Jr: Experience-dependent regulation of Leu-enkephalin hydrolysis in rat plasma. Peptides 14:161-167, 1993
Ryan JW, Chung AYK, Nearing JA, Valido FA, Shun-Cun C, Berryer P: A radiochemical assay for aminopeptidase N. Anal Biochem 210:27-33, 1993
Lucius R, Sievers J, Mentlein R: Enkephalin metabolism by microglia aminopeptidase-N (CD13). J Neurochem 64:1814-1847, 1995
Thornton JR, Losowsky MS: Opioid peptides and primary biliary cirrhosis. BMJ 297:1501-1504, 1988
Thornton JR, Dean H, Losowsky MS: Is ascites caused by impaired hepatic inactivation of blood borne endogenous opioid peptides? Gut 29:1167-1172, 1988
Thornton JR, Losowsky MS: Methionine enkephalin is increased in plasma in acute liver disease and is present in bile and urine. J Hepatol 8:53-59, 1989
Thornton JR, Losowsky MS: Plasma leucine enkephalin is increased in liver diseases. Gut 30:1392-1395, 1989
Swain MG, Rothman RB, Xu H, Vergalla J, Begasa NV, Jones EA: Endogenous opioids accumulate in plasma in rat model of acute cholestasis. Gastroenterology 103:630-635, 1992
Yurdaydin C, Li Y, Ha J-H, Jones EA, Rothman R, Basile AS: Brain and plasma levels of opioid peptides are altered in rats with thioace tamide-induced fulminant hepatic failure: Implications for the treatment of hepatic encephalopathy with opioid antagonists. J Pharmacol Exp Ther 273:185-192, 1995
Jones EA, Bergasa NV: The pruritus of cholestasis: From bile acids to opiate antagonists. Hepatology 5:884-887, 1990
Bergasa NV, Jones EA: The pruritus of cholestasis: potential pathogenic and therapeutic implications of opioids. Gastroenterology 108:1582-1588, 1995
Bergasa NV, Talbot TL, Alling DW, Schmitt JM, Walker EC, Bake r BL, Korenman JC, Park Y, Hoofnagle JH, Jones EA: A controlled trial of naloxone infusions for the pruritus of chronic cholestasis. Gastroenterology 102:544-549, 1992
Bergasa NV, Alling DW, Talbot TL, Swain MG, Yurdaydin C, Turner ML, Schmidt JM, Walker EC, Jones EA: Effects of naloxone infusions in patients with the pruritus of cholestasis. Ann Intern Med 123:161-167, 1995
Janas RM, Trette r J, Warnawin K, Pawlowska J, Rujner J, Socha J: Methionine enkephalin concentration and enkephalin-degrading activity are elevated in blood in children with cholestasis. Eur J Gastroenterol Hepatol 6:539-545, 1994
Janas RM, Tretter J, Warnawin K, Pawlowska J, Rujner J, Socha J: Methionine enkephalin and aminopeptidase-M activity in blood in children with cholestasis before and after treatment with ursodeoxycholic acid. In Cholestatic Liver Diseases. GP van Berge Henegouwen, J De Grote, B van Hoek, S Matern, RW Stockbrugger (eds). Dordrecht, Kluwer Academic Publishers, 1994, pp 226-235
Janas RM, Marks DL, LaRusso NF: An aluminium silicate binding assay for quantitation of degradation of cholecystokinin octapeptide and other short peptides. Anal Biochem 36:6-11, 1992
Xu Y, Wellner D, Schneilberg DA: Substance P and bradykinin are natural inhibitors of CD13/aminopeptidase-N. Biochem Biophys Res Commun 208:664-674, 1995
Janas RM: Serum aminopeptidase-M activity in cholestasis. Is the enzyme a marker or co-factor of the disease? Abstract Book: Inte rnational Falk Workshop, October 23-24, 1995 Basel, Switzerland, A-40
Rights and permissions
About this article
Cite this article
Janas, R.M., Socha, J., Warnawin, K. et al. Further Studies on Aminopeptidase-M in Blood in Children with Cholestatic Liver Diseases and Viral Hepatitis. Dig Dis Sci 44, 170–176 (1999). https://doi.org/10.1023/A:1026626822298
Issue Date:
DOI: https://doi.org/10.1023/A:1026626822298