Abstract
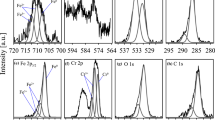
From the higher fraction of grain boundaries in nanocrystalline substances a different corrosion behaviour in comparison to the conventional polycrystalline material can be expected, which may be utilised for the development of new corrosion resistant alloys. Therefore, the oxidation behaviour of these two different crystallisation states of FeAl8 was compared by means of electrochemical and surface analytical experiments. The oxide films formed after electrochemical passivation were investigated by Auger Electron Spectroscopy. The application of inelastic peak shape analysis by the method of Tougaard showed, that for both materials the oxide layer may be described by a model of a (below the contamination) buried layer with a thickness of only a few nanometers depending on the preparation conditions. Factor Analysis was applied for the evaluation of the differentiated low energy Auger electron spectra (20–100 eV) as a function of depth profiling sputtering time. For both, the nanocrystalline and the polycrystalline material, the inner part of the oxide layer was enriched in Al, whereas the very outer part (surface region) was enriched in Fe. No differences concerning the sputtering time for removal of the oxide layers were found for the two alloys.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 6 September 1998 / Revised: 11 February 1999 / Accepted: 18 February 1999
Rights and permissions
About this article
Cite this article
John, A., Zeiger, W., Scharnweber, D. et al. Surface analytical investigation of the electrochemical and corrosion behaviour of nanocrystalline FeAl8. Fresenius J Anal Chem 365, 136–141 (1999). https://doi.org/10.1007/s002160051459
Issue Date:
DOI: https://doi.org/10.1007/s002160051459