Summary
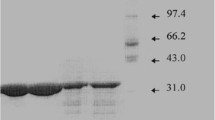
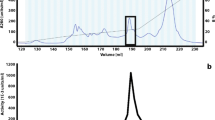
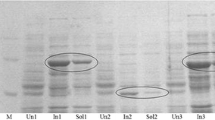
Anhydrotetracycline oxygenase was purified from cleared cell lysates ofStreptomyces rimosus, using Fast Protein Liquid Chromatography. The preparation was subjected to preparative gel electrophoresis, yielding a single major species which was sufficiently pure to allow direct N-terminal amino acid sequence determination.
Similar content being viewed by others
References
Behal, V., Hostalek, Z and Vanek, Z (1979) Biotechnol. Lett. 1, 177–182.
Binnie, C., Warren, M. and Butler, M.J. (1989) J. Bact. 171, 887–895.
Butler, M.J., Friend, E.J., Hunter, I.S., Sugden, D.A. and Warren, M. (1989) Molec. Gen. Genet. 215, 231–238.
Rhodes, P.M., Winskill, N., Friend, E.J. and Warren M (1981) J. Gen Microbiol. 124, 329–338.
Vancurova, I., Volc, J., Flieger, M., Neuzil, J., Novotna, J., Vlach, J. and Behal, V. (1988) Biochem. J. 253, 263–267.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Butler, M.J., Gedge, B.N. Purification of anhydrotetracycline oxygenase fromStreptomyces rimosus using fast protein liquid chromatography. Biotechnol Tech 3, 235–238 (1989). https://doi.org/10.1007/BF01876055
Issue Date:
DOI: https://doi.org/10.1007/BF01876055