Abstract
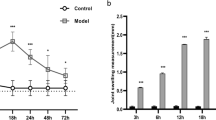
Casticin, a flavonoid isolated from Vitex trifolia, has been shown to have anti-inflammatory and antitumor effects in previous studies. Osteoarthritis (OA) is a disease based on degenerative pathological changes. The disease process is often accompanied by inflammatory pathological changes. However, there is no safe and effective drug for prevention and treatment. In the present study, we aimed to clarify the role of casticin in the murine model of destabilization of the medial meniscus (DMM). Male BALB/c mice were randomly divided into three groups: Sham, DMM-induced OA treated with vehicle, and DMM-induced OA treated with casticin. Our results indicated that the casticin treatments markedly reduced the destruction of cartilage and OARSI grades compared with those of the vehicle-treated mice. The levels of matrix metalloproteinase-13 (MMP13) in cartilage were also significantly reduced in the casticin-treated mice. Casticin also significantly regulated oxidative stress and reduced inflammation in the cartilage of mice with OA. These results suggest that casticin prevents the development of posttraumatic OA in mice. Consequently, decreased reactive oxygen species levels and suppressed proinflammatory cytokine production were confirmed in casticin-treated IL-1β-stimulated ADTC5 cells. After casticin treatment, the NF-κB signaling pathway was significantly inhibited in the cells. It can be concluded that casticin can alleviate arthritis-related cartilage degeneration by inhibiting ROS-mediated NF-κB signaling pathway in vitro and in vivo.






Similar content being viewed by others
References
Hunter, D.J., and S. Bierma-Zeinstra. 2019. Osteoarthritis. Lancet (London, England) 393:1745–1759.
Dieppe, P.A., and L.S. Lohmander. 2005. Pathogenesis and management of pain in osteoarthritis. Lancet (London, England) 365:965–973.
Suri, P., D.C. Morgenroth, and D.J. Hunter. 2012. Epidemiology of osteoarthritis and associated comorbidities. PM & R : The Journal of Injury, Function, and Rehabilitation 4:s10–s19.
Loeser, R.F., S.R. Goldring, C.R. Scanzello, and M.B. Goldring. 2012. Osteoarthritis: A disease of the joint as an organ. Arthritis and Rheumatism 64: 1697–1707.
Zeng, C., J. Wei, Persson MSM, A. Sarmanova, M. Doherty, D. Xie, Y. Wang, X. Li, J. Li, H. Long, et al. 2018. Relative efficacy and safety of topical non-steroidal anti-inflammatory drugs for osteoarthritis: A systematic review and network meta-analysis of randomised controlled trials and observational studies. British Journal of Sports Medicine 52: 642–650.
Lazzaroni, M., and G. Bianchi Porro. 2004. Gastrointestinal side-effects of traditional non-steroidal anti-inflammatory drugs and new formulations. Alimentary Pharmacology & Therapeutics 20: 48–58.
Philp, A.M., E.T. Davis, and S.W. Jones. 2017. Developing anti-inflammatory therapeutics for patients with osteoarthritis. Rheumatology (Oxford) 56: 869–881.
Lieberthal, J., N. Sambamurthy, and C.R. Scanzello. 2015. Inflammation in joint injury and post-traumatic osteoarthritis. Osteoarthritis and Cartilage 23: 1825–1834.
Goldring, M.B., M. Otero, K. Tsuchimochi, K. Ijiri, and Y. Li. 2008. Defining the roles of inflammatory and anabolic cytokines in cartilage metabolism. Annals of the Rheumatic Diseases 67: 75–82.
Kapoor, M., J. Martel-Pelletier, D. Lajeunesse, J.P. Pelletier, and H. Fahmi. 2011. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nature Reviews Rheumatology 7: 33–42.
Wojdasiewicz, P., Ł.A. Poniatowski, and D. Szukiewicz. 2014. The role of inflammatory and anti-inflammatory cytokines in the pathogenesis of osteoarthritis. Mediators of Inflammation 2014: 561459.
Prasadam, I., R. Crawford, and Y. Xiao. 2012. Aggravation of ADAMTS and matrix metalloproteinase production and role of ERK1/2 pathway in the interaction of osteoarthritic subchondral bone osteoblasts and articular cartilage chondrocytes -- possible pathogenic role in osteoarthritis. The Journal of Rheumatology 39: 621–634.
Mengshol, J.A., M.P. Vincenti, C.I. Coon, A. Barchowsky, and C.E. Brinckerhoff. 2000. Interleukin-1 induction of collagenase 3 (matrix metalloproteinase 13) gene expression in chondrocytes requires p38, c-Jun N-terminal kinase, and nuclear factor κB: Differential regulation of collagenase 1 and collagenase 3. Arthritis and Rheumatism 43: 801.
Kobayakawa, J., F. Sato-Nishimori, M. Moriyasu, and Y. Matsukawa. 2004. G2-M arrest and antimitotic activity mediated by casticin, a flavonoid isolated from Viticis Fructus (Vitex rotundifolia Linne fil.). Cancer Letters 208: 59–64.
Ling, Y., J. Zhu, M. Fan, B. Wu, L. Qin, and C. Huang. 2012. Metabolism studies of casticin in rats using HPLC-ESI-MS(n). Biomedical Chromatography 26: 1502–1508.
Haïdara, K., L. Zamir, Q. Shi, and G. Batist. 2006. The flavonoid Casticin has multiple mechanisms of tumor cytotoxicity action. Cancer Letters 242: 180–190.
Liou, C.J., C.Y. Cheng, K.W. Yeh, Y.H. Wu, and W.C. Huang. 2018. Protective effects of Casticin from Vitex trifolia alleviate Eosinophilic airway inflammation and oxidative stress in a murine asthma model. Frontiers in Pharmacology 9: 635.
Liou, C.J., W.B. Len, S.J. Wu, C.F. Lin, X.L. Wu, and W.C. Huang. 2014. Casticin inhibits COX-2 and iNOS expression via suppression of NF-kappaB and MAPK signaling in lipopolysaccharide-stimulated mouse macrophages. Journal of Ethnopharmacology 158 (Pt a): 310–316.
Lee, H., K.H. Jung, H. Lee, S. Park, W. Choi, and H. Bae. 2015. Casticin, an active compound isolated from Vitex Fructus, ameliorates the cigarette smoke-induced acute lung inflammatory response in a murine model. International Immunopharmacology 28: 1097–1101.
Mesaik, M.A., S. Murad, K.M. Khan, R.B. Tareen, A. Ahmed, and M.I. Choudhary. 2009. Isolation and immunomodulatory properties of a flavonoid, casticin from Vitex agnus-castus. Phytotherapy Research 23: 1516–1520.
Glasson, S.S., T.J. Blanchet, and E.A. Morris. 2007. The surgical destabilization of the medial meniscus (DMM) model of osteoarthritis in the 129/SvEv mouse. Osteoarthritis and Cartilage 15: 1061–1069.
Glasson, S.S., M.G. Chambers, W.B. Van Den Berg, and C.B. Little. 2010. The OARSI histopathology initiative - recommendations for histological assessments of osteoarthritis in the mouse. Osteoarthritis and Cartilage 18 (Suppl 3): S17–S23.
Vinatier, C., C. Merceron, and J. Guicheux. 2016. Osteoarthritis: From pathogenic mechanisms and recent clinical developments to novel prospective therapeutic options. Drug Discovery Today 21: 1932–1937.
Kuo, C.F., J.D. Su, C.H. Chiu, C.C. Peng, C.H. Chang, T.Y. Sung, S.H. Huang, W.C. Lee, and C.C. Chyau. 2011. Anti-inflammatory effects of supercritical carbon dioxide extract and its isolated carnosic acid from Rosmarinus officinalis leaves. Journal of Agricultural and Food Chemistry 59: 3674–3685.
Latourte, A., C. Cherifi, J. Maillet, H.K. Ea, W. Bouaziz, T. Funck-Brentano, M. Cohen-Solal, E. Hay, and P. Richette. 2017. Systemic inhibition of IL-6/Stat3 signalling protects against experimental osteoarthritis. Annals of the Rheumatic Diseases 76: 748–755.
Zhao, Y., Y. Li, R. Qu, X. Chen, W. Wang, C. Qiu, B. Liu, X. Pan, L. Liu, K. Vasilev, J. Hayball, S. Dong, and W. Li. 2019. Cortistatin binds to TNF-alpha receptors and protects against osteoarthritis. EBioMedicine 41: 556–570.
Latimer, N., J. Lord, R.L. Grant, R. O'Mahony, J. Dickson, and P.G. Conaghan. 2009. Cost effectiveness of COX 2 selective inhibitors and traditional NSAIDs alone or in combination with a proton pump inhibitor for people with osteoarthritis. BMJ 339: b2538.
Wylie, J.D., J.C. Ho, S. Singh, D.R. McCulloch, and S.S. Apte. 2012. Adamts5 (aggrecanase-2) is widely expressed in the mouse musculoskeletal system and is induced in specific regions of knee joint explants by inflammatory cytokines. Journal of Orthopaedic Research 30: 226–233.
Hitchon, C.A., and H.S. El-Gabalawy. 2004. Oxidation in rheumatoid arthritis. Arthritis Research & Therapy 6: 265–278.
Valko, M., D. Leibfritz, J. Moncol, M.T. Cronin, M. Mazur, and J. Telser. 2007. Free radicals and antioxidants in normal physiological functions and human disease. The International Journal of Biochemistry & Cell Biology 39: 44–84.
Griffiths, H.R. 2005. ROS as signalling molecules in T cells--evidence for abnormal redox signalling in the autoimmune disease, rheumatoid arthritis. Redox Report 10: 273–280.
Henrotin, Y.E., P. Bruckner, and J.P. Pujol. 2003. The role of reactive oxygen species in homeostasis and degradation of cartilage. Osteoarthritis and Cartilage 11: 747–755.
Kaminskyy, V.O., and B. Zhivotovsky. 2014. Free radicals in cross talk between autophagy and apoptosis. Antioxidants & Redox Signaling 21: 86–102.
Wang, Y., R. Branicky, A. Noe, and S. Hekimi. 2018. Superoxide dismutases: Dual roles in controlling ROS damage and regulating ROS signaling. The Journal of Cell Biology 217: 1915–1928.
Yang, C.S., J.J. Kim, S.J. Lee, J.H. Hwang, C.H. Lee, M.S. Lee, and E.K. Jo. 2013. TLR3-triggered reactive oxygen species contribute to inflammatory responses by activating signal transducer and activator of transcription-1. Journal of Immunology 190: 6368–6377.
Kim, J., M. Xu, R. Xo, A. Mates, G.L. Wilson, A.W. Pearsall, and V. Grishko. 2010. Mitochondrial DNA damage is involved in apoptosis caused by pro-inflammatory cytokines in human OA chondrocytes. Osteoarthritis and Cartilage 18: 424–432.
Glineur, C., E. Davioud-Charvet, and B. Vandenbunder. 2000. The conserved redox-sensitive cysteine residue of the DNA-binding region in the c-Rel protein is involved in the regulation of the phosphorylation of the protein. The Biochemical Journal 352: 583–591.
Janssen-Heininger, Y.M., M.E. Poynter, and P.A. Baeuerle. 2000. Recent advances towards understanding redox mechanisms in the activation of nuclear factor kappaB. Free Radical Biology & Medicine 28: 1317–1327.
Lai, Y., X. Bai, Y. Zhao, Q. Tian, B. Liu, E.A. Lin, Y. Chen, B. Lee, C.T. Appleton, F. Beier, X.P. Yu, and C.J. Liu. 2014. ADAMTS-7 forms a positive feedback loop with TNF-alpha in the pathogenesis of osteoarthritis. Annals of the Rheumatic Diseases 73: 1575–1584.
Minashima, T., K.A. Campbell, S.R. Hadley, Y. Zhang, and T. Kirsch. 2014. The role of ANK interactions with MYBBP1a and SPHK1 in catabolic events of articular chondrocytes. Osteoarthritis and Cartilage 22: 852–861.
Campbell, K.A., T. Minashima, Y. Zhang, S. Hadley, Y.J. Lee, J. Giovinazzo, M. Quirno, and T. Kirsch. 2013. Annexin A6 interacts with p65 and stimulates NF-kappaB activity and catabolic events in articular chondrocytes. Arthritis and Rheumatism 65: 3120–3129.
Yasuda, T. 2011. Activation of Akt leading to NF-κB up-regulation in chondrocytes stimulated with fibronectin fragment. Biomedical Research (Tokyo, Japan) 32: 209–215.
Zheng, W., Z. Tao, L. Cai, C. Chen, C. Zhang, Q. Wang, X. Ying, W. Hu, and H. Chen. 2017. Chrysin attenuates IL-1β-induced expression of inflammatory mediators by suppressing NF-κB in human osteoarthritis chondrocytes. Inflammation 40: 1143–1154.
Funding
These studies were supported by the National Natural Science Foundation of China. Award Number: 81672228 and 81702199.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data analysis were performed by Jun Chu, Tao Jiang and Bo Yan. Jun Chu, Jie Zhang and Zhenyu Zheng performed the experiments. The first draft of the manuscript was written by Zhongmin Zhang, and Lilan Peng. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Data Availability
The data generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chu, J., Yan, B., Zhang, J. et al. Casticin Attenuates Osteoarthritis-Related Cartilage Degeneration by Inhibiting the ROS-Mediated NF-κB Signaling Pathway in vitro and in vivo. Inflammation 43, 810–820 (2020). https://doi.org/10.1007/s10753-019-01167-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-019-01167-y