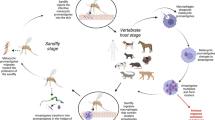
Abstract
Leishmaniasis is a zoonosis caused by protozoan species of the genus Leishmania. It generates different clinical manifestations in humans and animals, and it infects multiple hosts. Leishmania parasites are transmitted by sandfly vectors. The main objective of this systematic review was to identify the host, or reservoir animal species, of Leishmania spp., with the exception of domestic dogs, that were recorded in Brazil. This review included identification of diagnostic methods, and the species of protozoan circulating in the country. For this purpose, a literature search was conducted across index journals. This study covered the period from 2001 to 2021, and 124 studies were selected. Eleven orders possible hosts were identified, including 229 mammalian species. Perissodactyla had the highest number of infected individuals (30.69%, 925/3014), with the highest occurrence in horses. In Brazil, the most commonly infected species were found to be: horses, domestic cats, rodents, and marsupials. Bats, that were infected by one or more protozoan species, were identified as potential reservoirs of Leishmania spp. Molecular tests were the most commonly used diagnostic methods (94 studies). Many studies have detected Leishmania spp. (n = 1422): Leishmania (Leishmania) infantum (n = 705), Leishmania (Viannia) braziliensis (n = 319), and Leishmania (Leishmania) amazonensis (n = 141). Recognizing the species of animals involved in the epidemiology and biological cycle of the protozoan is important, as this allows for the identification of environmental biomarkers, knowledge of Leishmania species can improve the control zoonotic leishmaniasis.

Similar content being viewed by others
References
Achilles GR, Kautzmann RP, Chagas HDF, Pereira-Silva JW, Almeida JF, Fonseca FR, Ferreira da Silva MN, Pessoa FAC, Nava AFD, Ríos-Velásquez CM (2021) Presence of trypanosomatids, with emphasis on Leishmania, in rodentia and didelphimorphia mammals of a rural settlement in the central amazon region. Mem Inst Oswaldo Cruz 116. https://doi.org/10.1590/0074-02760200427
Afonso MMS, Gomes AC, Meneses CRV, Rangel EF (2005) Studies on the feeding habits of Lutzomyia (N.) intermedia (Diptera, Psychodidae), vector of cutaneous leishmaniasis in Brazil. Cad Saúde Pública 21:1816–1820. https://doi.org/10.1590/S0102-311X2005000600030
Aguilar CM, Rangel EF, Deane LM (1986) Cutaneous leishmaniasis is frequent in equines from an endemic area in Rio de Janeiro, Brazil. Mem Inst Oswaldo Cruz 81:471–472
Ahuir-Baraja AE, Ruiz MP, Garijo MM, Llobat L (2021) Feline leishmaniosis: an emerging public health problem. Vet Scienc 8:173. https://doi.org/10.3390/vetsci8090173
Alencar LO, Chagas FSC, Silva MAG, Santos HD (2020) Leishmaniose em Equinos do Município de Araguaína, Tocantins: Levantamento da Soroprevalência Através da Reação de Imunofluorescência Indireta (RIFI). DESAFIOS - Rev Interdiscip Univ Fed Tocantins 7:283–287. https://doi.org/10.20873/uftsupl2020-8634
Almeida JC, Melo RPB, Kim PCP, Guerra NR, Alves LC, Costa DF, Alves CJ, Porto WJN, Mota RA (2018) Molecular and serological investigation of infectious diseases in captive and free-range crab-eating fox (Cerdocyon thous - Linnaeus, 1776) from northeastern Brazil. Acta Parasitol 63:184–189. https://doi.org/10.1515/ap-2018-0021
Al-soud WA, Radstrom P (2001) Purificação e caracterização de componentes inibidores de PCR em células sanguíneas. J Clin Microbiol 39:485–493. https://doi.org/10.1128/JCM.39.2.485-493.2001
Araujo VAL, Boite MC, Cupolillo E, Jansen AM, Roque ALR (2013) Mixed infection in the anteater Tamandua tetradactyla (Mammalia: Pilosa) from Pará State, Brazil: Trypanosoma cruzi, T. rangeli and Leishmania Infantum. Parasitol 140:455–460. https://doi.org/10.1017/S0031182012001886
Baneth G, Koutinas AF, Solano-Gallego L, Bourdeau P, Ferrer L (2008) Canine leishmaniosis–new concepts and insights on an expanding zoonosis: part one. Trends Parasitol 24:324–330. https://doi.org/10.1016/j.pt.2008.04.001
Bañuls AL, Hide M, Prugnolle F (2007) Leishmania and the leishmaniases: a parasite genetic update and advances in taxonomy, epidemiology and pathogenicity in humans. Adv Parasitol 64:1–458. https://doi.org/10.1016/S0065-308X(06)64001-3
Basano SA, Camargo LMA (2004) Leishmaniose tegumentar americana: histórico, epidemiologia e perspectivas de controle. Rev Bras Epidemiol 7:328–337
Batista JF, Neto FDCRM, Lopes KSPDP, Sato MO, Costa CHN, de Mendonça IL (2020) Transmission of Leishmania infantum from cats to dogs. Rev Bras Parasitol Vet 29:1–11. https://doi.org/10.1590/S1984-29612020099
Berenguer LKAR, Gomes CFCA, Nascimento JO, Bernardi JCM, Lima VFS, Oliveira JB, Ramos CAN, Ramos RAN, Alves LC (2021) Leishmania infantum infection in a domestic cat: a real threat or an occasional finding? Acta Parasitol 66:673–676. https://doi.org/10.1007/s11686-020-00294-z
Bern C, Courtenay O, Alvar J (2010) Of cattle, sand flies and men: a systematic review of risk factor analyses for South Asian visceral leishmaniasis and implications for elimination. PLoS Negl Trop Dis 4(2):e599. https://doi.org/10.1371/journal.pntd.0000599
Berzunza-Cruz M, Rodríguez-Moreno Á, Gutiérrez-Granados G, González-Salazar C, Stephens CR, Hidalgo-Mihart M, Marina CF, Rebollar-Téllez EA, Bailón- Martínez D, Balcells CD, Ibarra-Cerdeña CN, Sánchez-Cordero V, Becker I (2015) Leishmania (L.) mexicana infected bats in Mexico: novel potential reservoirs. PLoS Negl Trop Dis 9(1):e0003438. https://doi.org/10.1371/journal.pntd.0003438
Bezerra JAB, De Medeiros Oliveira IVP, Yamakawa AC, Nilsson MG, Tomaz KLR, De Oliveira KDS, Da Rocha CS, Calabuig CIP, Fornazari F, Langoni H, De Paula Antunes JMA (2019) Serological and molecular investigation of Leishmania spp. Infection in cats from an area endemic for canine and human leishmaniasis in northeast Brazil. Rev Bras Parasitol Vet 28:790–796. https://doi.org/10.1590/s1984-29612019082
Brandão EMV, Xavier SCC, Rocha FL, Lima CFM, Candeias ÍZ, Lemos FG, Azevedo FC, Jansen AM, Roque ALR (2020) Wild and domestic canids and their interactions in the transmission cycles of Trypanosoma cruzi and Leishmania spp. in an area of the Brazilian Cerrado. Pathogens 9:1–19. https://doi.org/10.3390/pathogens9100818
Brandão-Filho OSP, Carvalho FG, Brito ME, Almeida FA, Nascimento LA (1994) American cutaneous leishmaniasis in Pernambuco, Brazil: eco-epidemiological aspects in “Zona da Mata” region. Mem Inst Oswaldo Cruz 89:445–449
Brandão-Filho SP, Brito ME, Carvalho FG, Ishikaw EA, Cupolillo E, Floeter-Winter L, Shaw JJ (2003) Wild and synanthropic hosts of Leishmania (Viannia) braziliensis in the endemic cutaneous leishmaniasis locality of Amaraji, Pernambuco State, Brazil. Trans R Soc Trop Med Hyg 97:291–296. https://doi.org/10.1016/S0035-9203(03)90146-5
Brasil (2005) Ministério da Saúde. Secretaria de Vigilância em Saúde. Guia de vigilância epidemiológica. - 6. ed.- Brasília: Ministério da Saúde. Available at http://portal.saude.gov.br/portal/arquivos/pdf/Guia_Vig_Epid_novo2.pdf. Accessed 02 Nov 2022
Brasil (2014) Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Manual de Vigilância e controle da leishmaniose visceral/ Ministério da Saúde, Secretaria de Vigilância em Saúde, Departamento de Vigilância Epidemiológica – 1. Ed., 5 reimpr. – Brasilia: ministério da Saúde. 120 p
Brasil (2016) Ministério da saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância das Doenças Transmissíveis. Manual de Vigilância, prevenção e controle de zoonoses: normas técnicas e operacionais. Brasília: Ministério da saúde. 121 p
Brazil RP, Nascimento MDSB, Macau RP (1987) Infecção natural do porco (Sus scrofa) por Leishmania em foco recente de leishmaniose tegumentar na Ilha de São Luiz, Maranhão. Mem Inst Oswaldo Cruz 82:145
Caldart ET, Freire RL, Ferreira FP, Ruffolo BB, Mareze M, Navarro IT (2017) Leishmania in synanthropic rodents (Rattus rattus): new evidence for the urbanization of Leishmania. Braz J Vet Parasitol 26:17–27. https://doi.org/10.1590/S1984-29612017001
Campbell-Lendrum D, Dujardin JP, Martinez E, Feliciangeli MD, Perez JE, de Silans LNM, Desjeux P (2001) Domestic and peridomestic transmission of American cutaneous leishmaniasis: changing epidemiological patterns present new control opportunities. Mem Inst Oswaldo Cruz 96:159–162. https://doi.org/10.1590/S0074-02762001000200004
Cândido SL, Pavelegini LAD, Pacheco TDA, Pacheco R de C, Silva VL de B, Morgado TO, Colodel EM, Nakazato L, Almeida A do BPF de, Dutra V (2021) Molecular detection of trypanosomatids in neotropical primates in the state of Mato Grosso, Midwest, Brazil. Rev Bras Parasitol Vet 30:e001321. https://doi.org/10.1590/S1984-29612021041
Cardoso RM, Araújo NNSL, Romero GAS, Souza TTCM, Dietrich AG, Mendes JD, Reis ML, Ferreira JBC, Hecht MM, Gurgel-Gonçalves R (2015) Expanding the knowledge about Leishmania species in wild mammals and dogs in the Brazilian savannah. Parasit Vectors 8:1–8. https://doi.org/10.1186/s13071-015-0780-y
Carneiro LA, Santos TV, Lima LVR, Ramos PKS, Campos MB, Silveira FT (2020) First report on feline leishmaniasis caused by Leishmania (Leishmania) amazonensis in Amazonian Brazil. Vet Parasitol Reg Stud Reports 19:100360. https://doi.org/10.1016/j.vprsr.2019.100360
Cássia-Pires R, Boité MC, D’Andrea PS, Herrera HM, Cupolillo E, Jansen AM, Roque ALR (2014) Distinct Leishmania species infecting wild caviomorph rodents (Rodentia: Hystricognathi) from Brazil. PLoS Negl Trop Dis 8. https://doi.org/10.1371/journal.pntd.0003389
Chagas E, Cunha AM, Ferreira LC, Deane L, Deane G, Guimarães FN, Paumgartten MJ, Sá B (1938) Leishmaniose visceral americana: relatório dos trabalhos realizados pela Comissão Encarregada do Estudo da Leishmaniose Visceral Americana em 1937. Mem Inst Oswaldo Cruz 33:89–229. https://doi.org/10.1590/S0074-02761938000100010
Conceição-Silva F, Alves CR (2014) Leishmanioses do Continente Americano. Rio de Janeiro: Ed. Fiocruz; 512 p
Costa-Val AP, Coura FM, Barbieri JM, Diniz L, Sampaio A, Reis JKP, Bueno BL, Gontijo CMF (2020) Serological study of feline leishmaniasis and molecular detection of Leishmania infantum and Leishmania braziliensis in cats (Felis catus). Braz J Vet Parasitol 29(2):e003520. https://doi.org/10.1590/S1984-29612020023
Escobar TA, Dowich G, Santos TP, Zuravski L, Duarte CA, Lübeck I, Manfredini V (2019) Assessment of Leishmania infantum infection in equine populations in a canine visceral leishmaniosis transmission area. BMC Vet Res 15:381. https://doi.org/10.1186/s12917-019-2108-1
Evers F, Ferreira FP, Navarro IT, Mitsuka-Breganó R, Pagliari S, Monica TC, Nino BSL, Freire RL (2017) Presence of anti-Leishmania spp. antibodies in slaughter horses in Brazil. Semin Cienc Agrar 38(6):3921–3926. https://doi.org/10.5433/1679-0359.2017v38n6p3921
Ferreira PRB, Larangeira DF, Oliveira LS, Malta MCC, Gomes MC, Bastos BL, Portela RW, Barrouin-Melo SM (2013) Teste de ELISA indireto para diagnóstico sorológico de leishmaniose visceral em canídeos silvestres. Pesq Vet Bras 33(4):528–534. https://doi.org/10.1590/S0100-736X2013000400018
Ferro C, Morrison AC, Torres M, Pardo R, Wilson ML, Tesh RB (1995) Age structure, blood-feeding behavior, and Leishmania chagasi infection in Lutzomyia longipalpis (Diptera: Psychodidae) atan endemic focus of visceral leishmaniasis in Colombia. J Med Entomol 32:618–629. https://doi.org/10.1093/jmedent/32.5.618
Fornazari F, Langoni H (2014) Principais zoonoses em mamíferos selvagens. Vet Zootec, 10–24. Available at: http://hdl.handle.net/11449/140902. Accessed 09 Dec 2021
Gómez-Hernández C, Bento EC, Rezende-Oliveira K, Nascentes GAN, Barbosa CG, Batista LR, Tiburcio MGS, Pedrosa AL, Lages-Silva E, Ramírez JD, Ramirez LE (2017) Leishmania infection in bats from a non-endemic region of Leishmaniasis in Brazil. Parasitol 144(14):1980–1986. https://doi.org/10.1017/S0031182017001500
Gontijo B, Carvalho MLR (2003) Leishmaniose tegumentar americana. Rev Soc Bras Med Trop 36(1):71–80. https://doi.org/10.1590/S0037-86822003000100011
Grimaldi G Jr, Tesh RB (1993) Leishmaniases of the New World: current concepts and implications for the future research. Clin Microbiol Rev 6(3):230–250. https://doi.org/10.1128/CMR.6.3.230
Hong A, Zampieri RA, Shaw JJ, Floeter-Winter LM, Laranjeira-Silva MF (2020) One health approach to Leishmaniases: understanding the disease dynamics through diagnostic tools. Pathog 9(10):809. https://doi.org/10.3390/pathogens9100809
Humberg RMP, Oshiro ET, Pires e Cruz MS, Ribolla PEM, Alonso DP, Ferreira AMT, Bonamigo RA, Tasso N Jr, Oliveira AG (2012) Short report: Leishmania chagasi in Opossums (Didelphis albiventris) in an urban area endemic for visceral leishmaniasis, Campo Grande, Mato Grosso do Sul, Brazil. Am J Trop Med Hyg 87(3):470–472. https://doi.org/10.4269/ajtmh.2012.11-0534
IBGE (2013) BIGS— Brazilian Institute of Geography and Statistics, 2013 [internet]. Result of the Census Data—2013. Avaible at https://www.agricultura.gov.br/assuntos/camaras-setoriais-tematicas/documentos/camaras-tematicas/insumos-agropecuarios/anosanterior/ibge-populacao-de-animais-de-estimacao-no-brasil-2013-abinpet-79.pdf/view>. Accessed 07 July 2021
Jones G, Teeling EC (2006) The evolution of echolocation in bats. Trends Ecol Evol 21(3):149–156. https://doi.org/10.1016/j.tree.2006.01.001
Khosravi S, Hejazi SH, Hashemzadeh M, Eslami G, Darani HY (2012) Molecular diagnosis of Old World leishmaniasis: Real-time PCR based on tryparedoxin peroxidase gene for the detection and identification of Leishmania spp. J Vector Borne Dis 49:15–18
Kuhls K, Keilonat L, Ochsenreither S, Schaar M, Schweynoch C, Presber W, Schönian G (2007) Multilocus microsatellite typing (MLMT) reveals genetically isolated populations between and within the main endemic regions of visceral leishmaniasis. Microbes Infect 9(3):334–343. https://doi.org/10.1016/j.micinf.2006.12.009
Lainson R, Shaw JJ (1987) Evolution, classification and geographical distribution. In: Peters W, Killick-Kendrick R (eds) The leishmaniases in biology and medicine. Academic Press, London, vol I, 12–120
Lampo M, Feliciangeli MD, Márquez LM, Bastidas C, Lau P (2000) A possible role of bats as a blood source for the Leishmania vector Lutzomyia longipalpis (Diptera: Psychodidae). Am J Trop Med Hyg 62(6):718–719. https://doi.org/10.4269/ajtmh.2000.62.718
Lopes EG, Sevá AP, Ferreira F, Nunes CM, Keid LB, Hiramoto RM, Ferreira HL, Oliveira TMFS, Bigotto MFD, Galvis-Ovallos F, Galati EAB, Soares RM (2017) Serological and molecular diagnostic tests for canine visceral leishmaniasis in Brazilian endemic area: one out of five seronegative dogs are infected. Epidemiol Infect 145(12):2436–2444. https://doi.org/10.1017/S0950268817001443
Luppi MM, Malta MCC, Silva TMA, Silva FL, Motta ROC, Miranda I, Ecco R, Santos RL (2008) Visceral leishmaniasis in captive wild canids in Brazil. Vet Parasitol 155(1–2):146–151. https://doi.org/10.1016/j.vetpar.2008.04.024
Luz E, Giovannoni M, Borba AM (1967) Infecção de Lutzomyia monticola por Leishmania enriettii. An Facul de Med Univ Fed do Paraná. Curitiba 9:121–128
Machado CJS, Silva EG, Vilani RM (2016) Use of an instrument of controversial public health policy: euthanasia of dogs contaminated by leishmaniasis in Brazil. Saúde Soc 25(1):247–258. https://doi.org/10.1590/S0104-12902016146918
Maia C, Campino L (2011) Can domestic cats be considered reservoir hosts of zoonotic leishmaniasis? Trends Parasitol 27(8):341–344. https://doi.org/10.1016/j.pt.2011.03.008
Malta MCC, Tinoco HP, Xavier MN, Vieira ALS, Costa EA, Santos RL (2010) Naturally acquired visceral leishmaniasis in non-human primates in Brazil. Vet Parasitol 169(1–2):193–197. https://doi.org/10.1016/j.vetpar.2009.12.016
Marfurt J, Nasereddin A, Niederwieser I, Jaffe CL, Beck H-P, Felger I (2003) Identification and differentiation of Leishmania species in clinical samples by PCR amplification of the miniexon sequence and subsequent restriction fragment length polymorphism analysis. J Clin Microbiol 41(7):3147–3153. https://doi.org/10.1128/JCM.41.7.3147-3153.2003
McFarlane RO, Sleigh A, McMichael T (2012) Synanthropy of wild mammals as a determinant of emerging infectious diseases in the Asian-Australasian Region. EcoHealth 9(1):24–35. https://doi.org/10.1007/s10393-012-0763-9
Medina H (1946–2001) Estudos sobre leishmaniose: I. Primeiros casos de leishmaniose espontânea observados em cobaios. Braz Arch Biol Technol 1:13–55. https://doi.org/10.1590/S1516-89132001000500003
Meneses CR, Azevedo AC, Costa SM, Costa WA, Rangel EF (2002) Ecology of American cutaneous leishmaniasis in the state of Rio de Janeiro. Brazil J Vector Ecol 27(2):207–214
Mol JPS, Soave SA, Turchetti AP, Pinheiro GRG, Pessanha AT, Malta MCC, Tinoco HP, Figueiredo LA, Gontijo NF, Paixão TA, Fujiwara RT, Santos RL (2015) Transmissibility of Leishmania infantum from maned wolves (Chrysocyon brachyurus) and bush dogs (Speothos venaticus) to Lutzomyia longipalpis. Vet Parasitol 212(3–4):86–91. https://doi.org/10.1016/j.vetpar.2015.08.024
Moraes-Silva E, Antunes FR, Rodrigues MS, Julião FS, Dias-Lima AG, Lemos-de-Sousa V, Alcantara AC, Reis EAG, Nakatani M, Badaró R, Reis MG, Pontes-de-Carvalho L, Franke CR (2006) Domestic swine in a visceral leishmaniasis endemic area produce antibodies against multiple Leishmania infantum antigens but apparently resist to L. infantum infection. Acta Trop 98(2):176–182. https://doi.org/10.1016/j.actatropica.2006.04.002
Morais RCS, Gonçalves SC, Costa PL, Silva KG, Silva FJ, Silva RP, Brito MEF, Brandão-Filho SP, Dantas-Torres F, Paiva-Cavalcanti M (2013) Detection of Leishmania infantum in animals and their ectoparasites by conventional PCR and real time PCR. Exp Appl Acarol 59:473–481. https://doi.org/10.1007/s10493-012-9611-4
Morais RCS, Oliveira CNC, Albuquerque SC, Silva LAMT, Pessoa-e-Silva R, Cruz HLA, Brito MEF, Cavalcanti MP (2016) Real-time PCR for Leishmania species identification: Evaluation and comparison with classical techniques. Exp Parasitol 165:43–50. https://doi.org/10.1016/j.exppara.2016.03.005
Moreira Júnior ED, Souza VMMD, Sreenivasan M, Lopes NL, Silva RBBD, Pontes-de-Carvalho LC (2003) Peridomestic risk factors for canine leishmaniasis in urban dwellings: new findings from a prospective study in Brazil. Am J Trop Med Hyg 69(4):393–397
Morrison AC, Ferro C, Tesh RB (1993) Host preferences of the sand fly Lutzomyia longipalpis at an endemic focus of American visceral leishmaniasis in Colombia. Am J Trop Med Hyg 49(1):68–75. https://doi.org/10.4269/ajtmh.1993.49.68
Ochsenreither S, Kuhls K, Schaar M, Presber W, Schonian G (2006) Multilocus microsatellite typing as a new tool for discrimination of Leishmania infantum MON-1 strains. J Clin Microbiol 44:495–503. https://doi.org/10.1128/JCM.44.2.495-503.2006
Oliveira FS, Pirmez C, Pires MQ, Brazil RP, Pacheco RS (2005) PCR-based diagnosis for detection of Leishmania in skin and blood of rodents from an endemic area of cutaneous and visceral leishmaniasis in Brazil. Vet Parasitol 129(3–4):219–227. https://doi.org/10.1016/j.vetpar.2005.01.005
Oliveira TM, Pereira VF, Benvenga GU, Martin MFA, Benassi JC, da Silva DT, Starke-Buzetti WA (2015a) Conjunctival swab PCR to detect Leishmania spp. in cats. Braz J Vet Parasitol 24:220–222. https://doi.org/10.1590/S1984-29612015016
Oliveira FM, Costa LHC, Barros TL, Ito PKRK, Colombo FA, Carvalho C, Pedro WA, Queiroz LH, Nunes CM (2015b) First detection of Leishmania spp. DNA in Brazilian bats captured strictly in urban areas. Acta Trop 150:176–181. https://doi.org/10.1016/j.actatropica.2015.07.010
Pan American Health Organization (PAHO) (2021) Leishmanioses: informe epidemiológico das Américas [Internet]. Num 10, dezembro de 2021. Washington, D.C.: OPAS. Available at: https://iris.paho.org/handle/10665.2/55386. Accessed 20 July 2022
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whitting P, Moher D (2021) The PRISMA (2020) statement: An updated guideline for reporting systematic reviews. Syst Rev 10:89. https://doi.org/10.1186/s13643-021-01626-4
Paranaíba LF, Pinheiro LJ, Torrecilhas AC, Macedo DH, Menezes-Neto A, Tafuri WL, Soares RP (2017) Leishmania enriettii (Muniz & Medina, 1948): A highly diverse parasite is here to stay. PLoS Pathog 13(5):e1006303. https://doi.org/10.1371/journal.ppat.1006303
Pedrassani D, Biolchi J, Gonçalves LR, Mendes NS, Zanatto DCS, Calchi AC, Machado RZ, André MR (2019) Molecular detection of vector-borne agents in cats in Southern Brazil. Braz J Vet Parasitol 28(4):632–643. https://doi.org/10.1590/s1984-29612019077
Pennisi MG, Persichetti MF (2018) Feline leishmaniosis: Is the cat a small dog? Vet Parasitol 251:131–137. https://doi.org/10.1016/j.vetpar.2018.01.012
Pita-Pereira D, Lins R, Oliveira MP, Lima RB, Pereira BA, Moreira OC, Brazil RP, Britto C (2012) SYBR Green-based Real-Time PCR targeting kinetoplast DNA can be used to discriminate between the main etiologic agents of Brazilian cutaneous and visceral leishmaniasis. Paras Vectors 12:5–1. https://doi.org/10.1186/1756-3305-5-15
Quinnell RJ, Courtenay O (2009) Transmission, reservoir host and control of zoonotic visceral leishmaniasis. Parasitol 136(14):1915–1934. https://doi.org/10.1017/S0031182009991156
Quintal APN, Ribeiro ES, Rodrigues FP, Rocha FS, Floeter-Winter LM, Nunes CM (2011) Leishmania spp. in Didelphis albiventris and Micoureus paraguayanus (Didelphimorphia: Didelphidae) of Brazil. Vet Parasitol 176(2–3):112–119. https://doi.org/10.1016/j.vetpar.2010.11.011
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing. Available at: https://www.R-project.org/. Accessed 24 Jul 2022
Riboldi E, Carvalho F, Romão PRT, Barcellos RB, Bello GL, Ramos RR, Oliveira RT, Araújo Júnior JP, Rossetti ML, Dallegrave E (2018) Molecular Method Confirms Canine Leishmania Infection Detected by Serological Methods in Non-Endemic Area of Brazil. Korean J Parasitol 56(1):11–19. https://doi.org/10.3347/kjp.2018.56.1.11
Sales KGS, Costa PL, Morais RCS, Otranto D, Brandão-Filho SP, Cavalcanti MP, Dantas-Torres F (2015) Identifcation of phlebotomine sand fly blood meals by real-time PCR. Parasit Vectors 8(1):230. https://doi.org/10.1186/s13071-015-0840-3
Santiago MEB, Vasconcelos RO, Fattori KR, Munari DP, Michelin AF, Lima VMF (2007) An investigation of Leishmania spp. in Didelphis spp. from urban and peri-urban areas in Bauru (São Paulo, Brazil). Vet Parasitol 150(4):283–290. https://doi.org/10.1016/j.vetpar.2007.09.026
Santis B, Santos EGOB, Cupolillo E, Porrozzi R, Cavalcanti AS, Santos BN, Moura ST, Machado K, Chaves SAM (2011) Characterization of Leishmania infantum species in dogs from the urban area of Cuiabá, State of Mato Grosso, Brazil. J Braz Soc Trop Med 44(6):771–773. https://doi.org/10.1590/S0037-86822011000600022
Santos NS, Pinho FA, Hlavac NRC, Nunes TL, Almeida NR, Solcà MS, Varjão BM, Portela RW, Rugani JN, Rêgo FD, Barrouin-Melo SM, Soares RP (2021) Feline Leishmaniasis caused by Leishmania infantum: parasite sequencing, seropositivity, and clinical characterization in an endemic area from Brazil. Front Vet Sci 8:734916. https://doi.org/10.3389/fvets.2021.734916
Savani ESMM, Almeida MF, Camargo MCGO, D’Auria SRN, Silva MMS, Oliveira ML, Sacramento D (2010) Detection of Leishmania (Leishmania) amazonensis and Leishmania (Leishmania) infantum chagasi in Brazilian bats. Vet Parasitol 168(1–2):5–10. https://doi.org/10.1016/j.vetpar.2009.10.019
Schallig HD, Silva ES, van der Meide WF, Schoone GJ, Gontijo CM (2007) Didelphis marsupialis (common opossum): A potential reservoir host for zoonotic leishmaniasis in the metropolitan region of Belo Horizonte (Minas Gerais, Brazil). Vector Borne Zoonotic Dis 7:387–393. https://doi.org/10.1089/vbz.2006.0651
Schönian G, Kuhls K, Mauricio IL (2011) Molecular approaches for a better understanding of the epidemiology and population genetics of Leishmania. Parasitol 138:405–425. https://doi.org/10.1017/S0031182010001538
Silva SM, Rabelo PFB, Gontijo NF, Ribeiro RR, Melo MN, Ribeiro VM, Michalick MSM (2010) First report of infection of Lutzomyia longipalpis by Leishmania (Leishmania) infantum from a naturally infected cat of Brazil. Vet Parasitol 174(1–2):150–154. https://doi.org/10.1016/j.vetpar.2010.08.005
Silva EM, Alves LC, Guerra NR, Farias MPO, Oliveira ELR, Cunha RCSC, Ramos RAN, Porto WJN (2016) Leishmania spp. in Didelphis spp. from northeastern Brazil. J Zoo Wildl Med 47(3):942–944. Available at https://www.jstor.org/stable/24773985. Accessed 01 Jan 2021
Silveira FT, Lainson R, Shaw JJ, Braga RR, Ishikawa EEA, Souza AAA (1991) Cutaneous leishmaniasis in the Amazon Region: isolation of Leishmania (Viannia) lainsoni from the rodent Agouti paca (Rodentia: Dasyproctidae) in Pará State, Brazil. Rev Inst Med Trop s Paulo 33(1):18–22. https://doi.org/10.1590/S0036-46651991000100004
Soares IR, Silva SO, Moreira FM, Prado LG, Fantini P, Maranhão RPA, Silva Filho JM, Melo MN, Palhares MS (2013) First evidence of autochthonous cases of Leishmania (Leishmania) infantum in horse (Equus caballus) in the Americas and mixed infection of Leishmania infantum and Leishmania (Viannia) braziliensis. Vet Parasitol 197(3–4):665–669. https://doi.org/10.1016/j.vetpar.2013.06.014
Soares MRA, Antunes JEL, de Mendonça IL, Lima RN, Costa CHN (2016) Occurrence of Lutzomyia longipalpis Lutz & Neiva 1912 and Cerdocyon thous Linnaeus 1977, in a visceral leishmaniasis endemic area in Brazil. Acta Trop 174:118–121. https://doi.org/10.1016/j.actatropica.2017.06.021
Sobrino R, Ferroglio E, Oleaga A, Romano A, Millan J, Revilla M, Arnal MC, Trisciuoglio A, Gortázar C (2008) Characterization of Widespread Canine Leishmaniasis among Wild Carnivores from Spain. Vet Parasitol 155(3–4):198–203. https://doi.org/10.1016/j.vetpar.2008.05.003
Soccol VT, Castro EA, Reifur L, Teixeira VN, Lange RR, Luz E (2021) Guinea pigs naturally infected by Leishmania enriettii: clinical analyses, parasite isolation and identification. Braz Arch Biol Technol 64:e21210095. https://doi.org/10.1590/1678-4324-75years-2021210095
Solano-Gallego L, Fernández-Bellon H, Morell P, Fondevila D, Alberola J, Ramis A, Ferrer L (2004) Histological and immunohistochemical study of clinically normal skin of Leishmania infantum- infected dogs. J Comp Pathol 130(1):7–12. https://doi.org/10.1016/s0021-9975(03)00063-x
Sousa KCM, Herrera HM, Domingos IH, Campos JBV, Santos IMC, Neves HH, Machado RZ, André MR (2014) Serological detection of Toxoplasma gondii, Leishmania infantum and Neospora caninum in cats from an area endemic for leishmaniasis in Brazil. Braz J Vet Parasitol 23(4):449–455. https://doi.org/10.1590/S1984-29612014078
Sousa SAP, Santos HD, Carvalho CA, Machado AM, Oliveira LE, Ribeiro TMP, Carreira AG, Galvão SR, Minharro S, Dias FEF, Jayme VS (2019) Acute visceral leishmaniasis in a domestic cat (Felis silvestris catus) from the state of Tocantins, Brazil. Semin Cienc Agrar 40(4):1723–1730. https://doi.org/10.5433/1679-0359.2019v40n4p1723
Souza AI, Barros EMS, Ishikawa E, Ilha IMN, Marin GRB, Nunes VLB (2005) Feline leishmaniasis due to Leishmania (Leishmania) amazonensis in Mato Grosso do Sul State, Brazil. Vet Parasitol 128(1–2):41–45. https://doi.org/10.1016/j.vetpar.2004.11.020
Souza AI, Nunes VLB, Borralho VM, Ishikawa EAY (2009) Domestic feline cutaneous leishmaniasis in the municipality of Ribas do Rio Pardo, Mato Grosso do Sul state, Brazil: a case report. J Venom Anim Toxins Incl Trop Dis 15(2):359–365. https://doi.org/10.1590/S1678-91992009000200017
Vale ECS, Furtado T (2005) Tegumentary leishmaniasis in Brazil: a historical review related to the origin, expansion and etiology. An Bras Dermatol 80(4):421–428. https://doi.org/10.1590/S0365-05962005000400015
Vedovello Filho D, Jorge FA, Lonardoni MVC, Teodoro U, Silveira TGV (2008) American cutaneous leishmaniasis in horses from endemic areas in the north-central Mesoregion of Paraná state, Brazil. Zoonoses Public Health 55(3):149–155. https://doi.org/10.1111/j.1863-2378.2008.01106.x
Vides JP, Schwardt TF, Sobrinho LSV, Marinho M, Laurenti MD, Biondo AW, Leutenegger C, Marcondes M (2011) Leishmania chagasi infection in cats with dermatologic lesions from an endemic area of visceral leishmaniosis in Brazil. Vet Parasitol 178(1–2):22–28. https://doi.org/10.1016/j.vetpar.2010.12.042
Vioti G, Leonel JAF, Lemes KM, Pereira VF, Ferreira HL, Keid LB, Madureira EH, Soares RM, Benassi JC, Oliveira TMFS (2019) Molecular detection of Leishmania spp. in cattle from Brazil by means of PCR using internal transcribed spacer 1. Braz J Vet Parasitol Vet 28(2):303–305. https://doi.org/10.1590/s1984-29612019003
WHO (2010) Control of the leishmaniases: report of a meeting of the WHO Expert Committee on the Control of Leishmaniases. WHO Press, Geneva, p 2010
WHO (2021) Leishmaniasis. Available at https://www.who.int/health-topics/leishmaniasis#tab=tab_1. Accessed 27 Jul 2022
WHO (2022) Leishmaniasis Facksheet. Available at https://www.who.int/en/news-room/fact-sheets/detail/leishmaniasis. Accessed 27 Oct 2022
Funding
This work was supported by the National Research and Development Council (CNPq) and Coordination for the Improvement of Higher Education Personnel (CAPES).
Author information
Authors and Affiliations
Contributions
Conceptualization and methodology: Fabiana Raquel Ratzlaff, Vanessa Osmari, Daniele da Silva, Jaíne Soares de Paula Vasconcellos, Luciana Pötter, Fagner D’ambroso Fernandes, José Américo de Mello Filho, Sônia de Avila Botton, Fernanda Silveira Flores Vogel, Luís Antônio Sangioni.
Writing—original draft preparation: Fabiana Raquel Ratzlaff, Luciana Pötter, Fagner D’ambroso Fernandes, Sônia de Avila Botton, Luís Antônio Sangioni.
Writing—review and editing: Fabiana Raquel Ratzlaff, Luciana Pötter, Fagner D’ambroso Fernandes, Sônia de Avila Botton, Luis Antônio Sangioni.
Corresponding author
Ethics declarations
Ethics approval
Not applicable
Consent to participate
Not applicable
Consent for publication
Not applicable
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Una Ryan
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
436_2023_7862_MOESM1_ESM.docx
Supplementary file1 Online Resource 1- Table 1: Studies carried out in Brazil between 2001 and 2021, identifying Leishmania species in animals belonging to different orders through molecular, serological and/or parasitological methods from collected organs and tissues (DOCX 44 KB)
436_2023_7862_MOESM2_ESM.docx
Supplementary file2 Online Resource 2- Fig. 2: Forest Plot indicating the total number of animals infected with the protozoan Leishmania spp. (0.25 = 25%) in relation to the total number surveyed (13,905 animals) selected by the authors in the systematic review (DOCX 19 KB)
436_2023_7862_MOESM3_ESM.docx
Supplementary file3 Online Resource 3- Fig. 3: Forest Plot indicating the frequency of domestic animals infected with one or more Leishmania species (0.26 = 26%) (DOCX 12 KB)
436_2023_7862_MOESM4_ESM.docx
Supplementary file4 Online Resource 4- Fig. 4: Forest Plot indicating the frequency of wild animals infected with one or more Leishmania species (0.24 = 24%) (DOCX 14 KB)
436_2023_7862_MOESM5_ESM.docx
Supplementary file5 Online Resource 5- Table 2: Description of the orders of mammals researched regarding the presence of the protozoan Leishmania spp., and the main host animal species where one or more species of the agent were identified (DOCX 20.8 KB)
436_2023_7862_MOESM6_ESM.docx
Supplementary file6 Online Resource 6- Fig. 5: Forest Plot indicating the total number of animals that showed clinical signs compatible with leismaniasis (0.11 = 11%) (DOCX 15 KB)
436_2023_7862_MOESM7_ESM.docx
Supplementary file7 Online Resource 7- Fig. 6: Forest Plot indicating the total number of domestic animals that showed clinical signs compatible with infection by Leishmania spp. in relation to the total number of domestic animals infected by the protozoan (0.14 = 14%) (DOCX 12 KB)
436_2023_7862_MOESM8_ESM.docx
Supplementary file8 Online Resource 8- Fig. 7: Forest Plot indicating the total number of wild animals that showed clinical signs compatible with infection by Leishmania spp. in relation to the total number of wild animals infected by the protozoan (0.06 = 6%) (DOCX 12 KB)

436_2023_7862_Fig2_ESM.png
Supplementary file9 Online Resource 9- Fig. 8: Number of mammals infected with Leishmania species, demonstrating the percentage of protozoan species circulating in the states of Amazonas, Pará and Tocantins in the North region of Brazil (PNG 5389 kb)

436_2023_7862_Fig3_ESM.png
Supplementary file10 Online Resource 10- Fig. 9: Number of mammals infected with Leishmania species, demonstrating the percentage of protozoan species circulating in the states of Pernambuco, Piauí, Bahia, Maranhão, Rio Grande do Norte, Paraíba and Sergipe in the Northeast region of Brazil (PNG 5389 kb)

436_2023_7862_Fig4_ESM.png
Supplementary file11 Online Resource 11- Fig. 10: Number of mammals infected with Leishmania species, demonstrating the percentage of protozoan species circulating in the states of São Paulo, Minas Gerais, Rio de Janeiro and Espírito Santo in the Southeast region of Brazil (PNG 5758 kb)

436_2023_7862_Fig5_ESM.png
Supplementary file12 Online Resource 12- Fig. 11 Number of mammals infected with Leishmania species, demonstrating the percentage of protozoan species circulating in the states of Mato Grosso, Mato Grosso do Sul, Goiás and Distrito Federal in the Midwest region of Brazil (PNG 5569 kb)

436_2023_7862_Fig6_ESM.png
Supplementary file13 Online Resource 13- Fig. 12 Number of mammals infected with Leishmania species, demonstrating the percentage of protozoan species circulating in the states of Paraná, Santa Catarina and Rio Grande do Sul in the South region of Brazil (PNG 5567 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ratzlaff, F.R., Osmari, V., da Silva, D. et al. Identification of infection by Leishmania spp. in wild and domestic animals in Brazil: a systematic review with meta-analysis (2001–2021). Parasitol Res 122, 1605–1619 (2023). https://doi.org/10.1007/s00436-023-07862-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-023-07862-y