Abstract
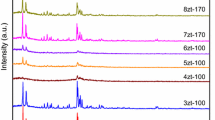
Micro-mesoporous zeolite materials differing in their content of micro- and mesopores are obtained by the recrystallization of modernite zeolite. Using physicochemical methods such as scanning electron microscopy, transmission electron microscopy, X-ray powder diffraction analysis, small-angle X-ray scattering analysis, low-temperature adsorption of nitrogen, temperature-programmed desorption of NH3, and IR spectroscopy, it is shown that recrystallization leads first to the formation of mesopores with sizes of 20–30 Å in zeolite crystals, then to the appearance of zeolite/MCM-41 nanocomposite, and finally to the complete conversion of zeolite into mesoporous MCM-41. During this process, the concentration of strong Brønsted acid sites accessible to pyridine bases first increases then drops. The catalytic properties of the synthesized materials are studied in the reaction of alkylation of benzene by dodecene-1. It is shown that the creation of transport pores and the increase in the accessibility of acid sites due to recrystallization under soft conditions facilitate the alkylation reaction.
Similar content being viewed by others
References
R. Dutartre, L. C. Menorva, et al., Micropor. Mater. 5–6, 311 (1996).
H. t Ajo, J. F. J. Lynch, F. Raatz, et al., Stud. Surf. Sci. Cat 62, 583 (1991).
D. McQueen and B. H. Chiche, et al., J. Catal. 161, 587 (1996).
Y. Tao, H. Kanoh, L. Abrams, et al., Chem. Rev. 106, 896 (2006).
C. H. Jacobsen, C. Madsen, J. Houzvicka, et al., J. Am. Chem. Soc. 122, 7116 (2000).
A. Karlsson and R. Schmidt, Micropor. Mesopor. Mater. 27, 181 (1999).
K. R. Kloestra, H. van Bekkum, and J. C. Jansen, Chem. Commun., p. 2281 (1997).
D. T. On and S. Kaliagine, Angew. Chem., Int. Ed. 40, 3248 (2001).
D. T. On, D. Lutic, and S. Kaliagine, Micropor. Mesopor. Mater. 44, 435 (2001).
I. I. Ivanova, A. S. Kuznetsov, V. V. Yuschenko, and E. E. Knyazeva, Pure Appl. Chem. 76, 1647 (2004).
S. Inagaki, M. Ogura, T. Inami, et al., Micropor. Mesopor. Mater. 74, 163 (2004).
I. I. Ivanova, A. S. Kuznetsov, O. A. Ponomareva, et al., Stud. Surf. Sci. Catal. 158, 121 (2005).
I. I. Ivanova and E. E. Knyazeva, RF Patent No. 2282587 (2006).
S. V. Konnov, Yu. V. Monakhova, E. E. Knyazeva, et al., Pet. Chem. 49, 79 (2009).
I. I. Ivanova, O. A. Ponomareva, E. E. Knyazeva, et al., RF Patent No. 2288034 (2006).
I. I. Ivanova, V. V. Ordomskii, E. E. Knyazeva, et al., RF Patent No. 2320631 (2008).
J. A. Kogal, B. V. Vora, and T. Imai, Appl. Catal. A: Gen. 221, 295 (2001).
C. Perego and P. Ingallina, Catal. Today 73, 3 (2002).
A. Galarneau, D. Desplantier, R. Dutartre, and F. Di Renzo, Micropor. Mesopor. Mater 27, 297 (1999).
T.-Ch. Tsai, I. Wang, and J.-Y. Liu, Green Chem. 5, 404 (2003).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.A. Ponomareva, S.E. Timoshin, E.E. Knyazeva, V.V. Ordomskii, V.V. Yushchenko, N.S. Kulikov, V.I. Zaikovskii, I.I. Ivanova, 2011, published in Zhurnal Fizicheskoi Khimii, 2011, Vol. 85, No. 12, pp. 2253–2259.
Rights and permissions
About this article
Cite this article
Ponomareva, O.A., Timoshin, S.E., Knyazeva, E.E. et al. Physicochemical and catalytic properties of micro-mesoporous zeolite materials. Russ. J. Phys. Chem. 85, 2103–2108 (2011). https://doi.org/10.1134/S0036024411120260
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024411120260