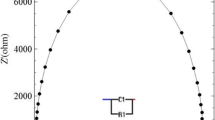
Abstract
The feasibility of electrolytic conductivity is performed using two-electrode system. A precision LCR meter is utilized to measure the ac resistance of electrolytic solution which is traceable to reference standard of impedance at CSIR-National Physical Laboratory. KCl solutions with different molality are used to establish the measurement system. The measurement of ac resistance is performed at frequencies above 10 kHz to minimize the impact of electrode polarization. Initially, the measurement setup was validated using a certified reference material. The KCl solutions with different molality are prepared gravimetrically. Thereafter, electrolytic conductivity of the aqueous solutions is computed for different molality of KCl solutions. The preliminary measurement suggested that the two-electrode system may be utilized for the measurement of electrolytic conductivity from 0.005 to 0.1 M upto 1 MHz.


Similar content being viewed by others
References
A. C. Lynch, A. E. Drake, and C. H. Dix, “Measurement of eddy-current conductivity, IEE Proc. A Phys. Sci. Meas. Instrum. Manag. Educ. Rev., (1983).
L. Callegaro, F. Durbiano, E. Orru and B. Trinchera, An impedance spectrometer for the metrology of electrolytic conductivity. IEEE Trans. Instrum. Meas., 62 (2013) 1766–1770.
P. Arquint, P. Arquint, H. Bühler and S. Worbs, Traceable standards for electrolytic conductivity measurements required for pharmaceutical production. Chimia (Aarau), 63 (2009) 647–649.
H. Czichos, T. Saito, and L. Smith, Handbook of metrology and testing. (2011).
R. H. Shreiner and K. W. Pratt, Primary standards and standard reference materials for electrolytic conductivity. (2004).
T. Asakai, I. Maksimov, S. Onuma, T. Suzuki, T. Miura and A. Hioki, New Japanese certified reference materials for electrolytic conductivity measurements. Accredit. Qual. Assur., 22 (2017) 73–81.
F. B. Gonzaga et al., CCQM–K36.2016 Electrolytic Conductivity at 0.5 S m-1 and 5 mS m-1 Final, (2015)
B. Magnusson, Report of key comparison CCQM–K92, 2013.
S.L. Schiefelbein, N.A. Fried, K.G. Rhoads and D.R. Sadoway, A high-accuracy, calibration-free technique for measuring the electrical conductivity of liquids. Rev. Sci. Instrum., 69 (1998) 3308–3313.
E. Yeager, A. J. Salkind, and F. R. Foulkes, Techniques of electrochemistry, J. Electrochem. Soc., (1974).
Agilent Technologies, Agilent impedance measurement handbook, 4th ed. 2009.
K.W. Pratt, W.F. Koch, Y.C. Wu and P.A. Berezansky, Molality-based primary standards of electrolytic conductivity (IUPAC technical report). Pure Appl. Chem., 73 (2001) 1783–1793.
Satish, S. Kumar, Babita, T. John and A.K. Saxena, Realization of coaxial reference air-lines as high frequency capacitance standard at CSIR-NPL. Measurement, 92 (2016) 166–171.
Satish, Babita, B. Khurana, S. Kumar and A.K. Saxena, Evaluation of four-terminal-pair capacitance standards using electrical equivalent circuit model. Measurement, 73 (2015) 121–126.
Satish, S. Kumar, Babita, T. John, and A. K. Saxena, Evaluation of air dielectric four-terminal-pair capacitance standards using resonance frequency of impedance elements, Measurement, 100 (2017) 176–182.
S. Singh, S. Kumar, Babita and T. John, Realization of four-terminal-pair capacitors as reference standards of impedance at high frequency using impedance-matrix method. IEEE Trans. Instrum. Meas., 66 (2017) 2129–2135.
Keysight, Operation and service manual: 16452A liquid test fixture.
Joint Committee For Guides In Metrology, “Evaluation of measurement data–Guide to the expression of uncertainty in measurement,” Int. Organ. Stand. Geneva ISBN, 50 (2008) 134.
Acknowledgements
Authors would like to thank Director CSIR-NPL for his constant support and encouragement to carry out this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Satish, Hemavathi, A., Krishna, A. et al. Feasibility of Electrolytic Conductivity of Aqueous Solutions Using Two-Electrode System. MAPAN 38, 337–341 (2023). https://doi.org/10.1007/s12647-022-00612-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12647-022-00612-y