Abstract
In the present study, a series of multipotent antioxidants (MPAOs), namely sulfur-containing BHT (S-BHT), derivatives were rationally designed and synthesized, and their inhibitory activities against free radicals and human cancer cell lines, HT29 (colon cancer) and MCF7 (breast cancer) were further evaluated. The experimental results showed that the Six out-of-eight S-BHT compounds had excellent antioxidant activity against DPPH radical with major enhancement compared to BHT. Among them, compounds 2b, 2a and 3b attained over 45% lower IC50 values than BHT. In vitro cytotoxicity, MTT assay was carried out using two human cancer cell lines, HT29 (colon cancer) and MCF7 (breast cancer) in addition to their non-tumorigenic counterparts to explore selectivity. In line with antioxidant activity, compounds 2a and 2b displayed the highest cytotoxicity effect on both cancer types. Interestingly, 2b not only exhibited superior cancer inhibition but also scored high selectivity index (SI = 5.2, 12.5) in colon and breast tissues, respectively, exceeding that of the standard chemotherapeutic drugs used 5-Fluorouracil (5-FU) and Tamoxifen (Tmx), with lower IC50 values. The results indicated that the symmetric S-BHT derivatives were significantly enhanced by the antioxidant potency and their ability as useful and promising selective anticancer agents.
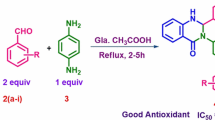
Graphic abstract
A series of sulfur-containing phenols (S-BHT) as multipotent antioxidants structure have been rationally designed and synthesized by the combination of the antiradical activity of the most well-known phenol moiety, butylated hydroxytoluene (BHT, primary antioxidant) with the antiperoxide (secondary antioxidant) and anti-proliferative fragments, sulfur-containing groups into one structure aiming to enhance scavenging ability and antiproliferative activity of the radicals. Accordingly, the bioactivity of S-BHT was analysed by antioxidant and anticancer in vitro studies and demonstrated remarkable cancer inhibition rates along with significant cytotoxicity and selectivity against HT29 and MCF7 cancer cell lines. This work efficiently sets the ground for eminent sulfur-containing phenols developed based on a well-known standard antioxidant BHT, with features superior to common drawbacks faced in cancer therapeutic approaches, in addition to the great potential holding as selective and biocompatible agent for cancer applications.









Similar content being viewed by others
References
May M 2014 Attacking an epidemic Nature 509 S50–S51
DeSantis C, Ma J, Bryan L and Jemal A 2014 Breast cancer statistics, 2013 CA Cancer J. Clin. 64 52
Kanchana A and Balakrishnan M 2011 Anti-cancer effect of saponins isolated from solanum trilobatum leaf extract and induction of apoptosis in human larynx cancer cell lines Int. J. Pharm. Pharm. Sci. 3 356
Ariffin A, Rahman N A, Yehye W A, Alhadi A A and Kadir F A 2014 PASS-assisted design, synthesis and antioxidant evaluation of new butylated hydroxytoluene derivatives Eur. J. Med. Chem. 87 564
Conner E M and Grisham M B 1996 Inflammation, free radicals, and antioxidants Nutrition 12 274
Schoneich C 1999 Reactive oxygen species and biological aging: A mechanistic approach Exp. Gerontol. 34 19
Sahinoglu T, Stevens C R, Bhatt B and Blake D R 1996 The role of reactive oxygen species in inflammatory disease: Evaluation of methodology Methods 9 628
Mills R and Wu G Z 2004 Synthesis and evaluation of novel prodrugs of foscarnet and dideoxycytidine with a universal carrier compound comprising a chemiluminescent and a photochromic conjugate J. Pharm. Sci. 93 1320
Ames B N, Gold L S and Willett W C 1995 The causes and prevention of cancer Proc. Natl. Acad. Sci. USA 92 5258
Rajeshkumar S 2016 Anticancer activity of eco-friendly gold nanoparticles against lung and liver cancer cells Genet. Eng. Biotechnol. J. 14 195
Solomon V R, Hu C and Lee H 2010 Design and synthesis of anti-breast cancer agents from 4-piperazinylquinoline: A hybrid pharmacophore approach Bioorg. Med. Chem. 18 1563
Chinery R, Brockman J A, Peeler M O, Shyr Y, Beauchamp R D and Coffey R J 1997 Antioxidants enhance the cytotoxicity of chemotherapeutic agents in colorectal cancer: a p53-independent induction of p21WAF1/CIP1 via C/EBPbeta Nat. Med. 3 1233
Lobo V, Patil A, Phatak A and Chandra N 2010 Free radicals, antioxidants and functional foods: Impact on human health Pharmacogn. Rev. 4 118
Stillson G H 1947 Alkylation of phenols U.S. Patent 2428745 A
Hilton J W 1989 Antioxidants: Function, types and necessity of inclusion in pet foods Can. Vet. J. 30 682
Botterweck A A M, Verhagen H, Goldbohm R A, Kleinjans J and van den Brandt P A 2000 Intake of butylated hydroxyanisole and butylated hydroxytoluene and stomach cancer risk: Results from analyses in the Netherlands cohort study Food Chem. Toxicol. 38 599
Yehye W A, Rahman N A, Alhadi A A, Khaledi H, Ng S W and Ariffin A 2012 Butylated hydroxytoluene analogs: Synthesis and evaluation of their multipotent antioxidant activities Molecules 17 7645
Barclay L R C, Vinqvist M R, Mukai K, Goto H, Hashimoto Y, Tokunaga A and Uno H 2000 On the antioxidant mechanism of curcumin: Classical methods are needed to determine antioxidant mechanism and activity Org. Lett. 2 2841
Erik K, Vladimír L and Cibulkova Z 2005 On the energetics of phenol antioxidants activity Pet. 47 33
Fukumoto L R and Mazza G 2000 Assessing antioxidant and prooxidant activities of phenolic compounds J. Agric. Food Chem. 48 3597
Bondet V, BrandWilliams W and Berset C 1997 Kinetics and mechanisms of antioxidant activity using the DPPH* free radical method Lebensm. Wiss. Technol. 30 609
De Gianni E and Fimognari C 2015 Anticancer mechanism of sulfur-containing compounds Enzymes 37 167
Cerella C, Dicato M, Jacob C and Diederich M 2011 Chemical properties and mechanisms determining the anti-cancer action of garlic-derived organic sulfur compounds Anticancer Agents Med. Chem. 11 267
Hong-Yu Z 2005 Structure-activity relationships and rational design strategies for radical-scavenging antioxidants Curr. Comput. Aided Drug Des. 1 257
Rzeski W, Matysiak J and Kandefer-Szerszen M 2007 Anticancer, neuroprotective activities and computational studies of 2-amino-1,3,4-thiadiazole based compound Bioorg. Med. Chem. 15 3201
Abdel-Rahman, T M 2006 Synthesis, reactions, and anticancer activity of some 1,3,4-thiadiazole/thiadiazine derivatives of carbazole Phosphorus Sulfur Silicon Relat. Elem. 181 1737
Vasoya S L, Paghdar D J, Chovatia P T and Joshi H S 2005 Synthesis of some new thiosemicarbazide and 1,3,4-thiadiazole heterocycles bearing benzo[b]thiophene nucleus as a potent antitubercular and antimicrobial agents J. Sci. I. R. Iran 16 33
Dawood K M and Gomha S M 2015 Synthesis and anti-cancer activity of 1,3,4-thiadiazole and 1,3-thiazole derivatives having 1,3,4-oxadiazole moiety J. Heterocycl. Chem. 52 1400
Aliabadi A, Eghbalian E and Kiani A 2013 Synthesis and evaluation of the cytotoxicity of a series of 1,3,4-thiadiazole based compounds as anticancer agents Iran. J. Basic Med. Sci. 16 1133
Gorinstein S, Martin-Belloso O, Katrich E, Lojek A, Ciz M, Gligelmo-Miguel N, Haruenkit R, Park Y S, Jung S T and Trakhtenberg S 2003 Comparison of the contents of the main biochemical compounds and the antioxidant activity of some Spanish olive oils as determined by four different radical scavenging tests J. Nutr. Biochem. 14 154
Mosmann T 1983 Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays J. Immunol. Met. 65 55
Badisa R B, Darling-Reed S F, Joseph P, Cooperwood J S, Latinwo L M and Goodman C B 2009 Selective cytotoxic activities of two novel synthetic drugs on human breast carcinoma MCF-7 cells Anticancer Res. 29 2993
Yehye W A, Rahman N A, Ariffin A, Hamid S B A, Alhadi A A, Kadir F A and Yaeghoobi M 2015 Understanding the chemistry behind the antioxidant activities of butylated hydroxytoluene (BHT): A review Eur. J. Med. Chem. 101 295
Wright J S, Johnson E R and DiLabio G A 2001 Predicting the activity of phenolic antioxidants: Theoretical method, analysis of substituent effects, and application to major families of antioxidants J. Am. Chem. Soc. 123 1173
Zhang H Y, Yang D P and Tang G Y 2006 Multipotent antioxidants: From screening to design Drug Discov. Today 11 749
Lucarini M, Pedulli G F and Cipollone M 1994 Bond-dissociation enthalpy of alpha-tocopherol and other phenolic antioxidants J. Org. Chem. 59 5063
Lim Y Y, Lim T T and Tee J J 2007 Antioxidant properties of several tropical fruits: A comparative study Food Chem. 103 1003
Henriquez C Bueno C Lissi E A and Encinas M V 2003 Thiols as chain transfer agents in free radical polymerization in aqueous solution Polymer 44 5559
Bordwell F G, Zhang X M, Satish A V and Cheng J P 1994 Assessment of the importance of changes in ground-state energies on the bond-dissociation enthalpies of the O-H bonds in phenols and the S-H Bonds in thiophenols J. Am. Chem. Soc. 116 6605
Wardman P and Vonsonntag C 1995 Kinetic factors that control the fate of thiyl radicals in cells Biothiols. Pt. A 251 31
Hermann R, Dey G R, Naumov S and Brede O 2000 Thiol radical cations and thiyl radicals as direct products of the free electron transfer from aromatic thiols to n-butyl chloride radical cations Phys. Chem. Chem. Phys. 2 1213
Eklund P C, Langvik O K, Warna J P, Salmi T O, Willfor S M and Sjoholm R E 2005 Chemical studies on antioxidant mechanisms and free radical scavenging properties of lignans Org. Biomol. Chem. 3 3336
Sharma O P and Bhat T K 2009 DPPH antioxidant assay revisited Food Chem. 113 1202
Ohkatsu Y, Haruna T and Osa T 1977 Kinetic evaluation of reactivity of phenolic derivatives as antioxidants for polypropylene J. Macromol. Sc. A 11 1975
Fujisawa S, Kadoma Y and Yokoe I 2004 Radical-scavenging activity of butylated hydroxytoluene (BHT) and its metabolites Chem. Phys. Lipids 130 189
Demirayak S, Benkli K and Guven K 2000 Synthesis and antimicrobial activities of some 3-arylamino-5-[2-(substituted 1-imidazolyl)ethyl]-1,2,4-triazole derivatives Eur. J. Med. Chem. 35 1037
Saito M, Sakagami H and Fujisawa S 2003 Cytotoxicity and apoptosis induction by butylated hydroxyanisole (BHA) and butylated hydroxytoluene (BHT) Anticancer Res. 23 4693
Abu N, Akhtar M N, Ho W Y, Yeap S K and Alitheen N B 2013 3-Bromo-1-hydroxy-9,10-anthraquinone (BHAQ) inhibits growth and migration of the human breast cancer cell lines MCF-7 and MDA-MB231 Molecules 18 10367
Yaacob N S, Kamal N N N M and Norazmi M N 2014 Synergistic anticancer effects of a bioactive subfraction of strobilanthes crispus and tamoxifen on MCF-7 and MDA-MB-231 human breast cancer cell lines BMC Complement. Altern. Med. 14 252
Etti I, Abdullah R, Hashim N M, Kadir A, Abdul A B, Etti C, Malami I, Waziri P and How C W 2016 Artonin E and structural analogs from artocarpus species abrogates estrogen receptor signaling in breast cancer Molecules 21 839
Anand M, Selvaraj V and Alagar M 2014 Synthesis, characterization and evaluation of antioxidant and anticancer activities of novel benzisoxazole-substituted-allyl derivatives Korean J. Chem. Eng. 31 659
Kan W L T, Yin C, Xu H X, Xu G, To K K W, Cho C H, Rudd J A and Lin G 2013 Antitumor effects of novel compound, guttiferone K, on colon cancer by p21Waf1/Cip1-mediated G0/G1 cell cycle arrest and apoptosis Int. J. Cancer 132 707
Acknowledgements
The authors wish to acknowledge the grant from the University of Malaya - Postgraduate Research Grant RP044C-17AET and PPP-2015B to conduct this study.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ahmad, M.H., Rahman, N.A., Kadir, F.A. et al. Design and synthesis of sulfur-containing butylated hydroxytoluene: antioxidant potency and selective anticancer agent. J Chem Sci 131, 107 (2019). https://doi.org/10.1007/s12039-019-1682-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-019-1682-x