Abstract
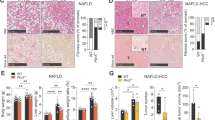
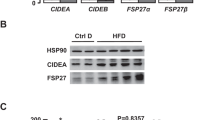
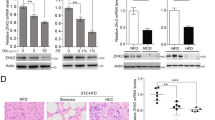
Annexin A6 (AnxA6) is a lipid-binding protein highly expressed in the liver, regulating cholesterol homeostasis and signaling pathways with a role in liver physiology. Here, we analyzed whether hepatic AnxA6 levels are affected by pathological conditions that are associated with liver dysfunction and liver injury. AnxA6 levels in the fatty liver of mice fed a high-fat diet, in ob/ob and db/db animals and in human fatty liver are comparable to controls. Similarly, AnxA6 levels appear unaffected in murine nonalcoholic steatohepatitis and human liver fibrosis. Accordingly, adiponectin, lysophosphatidylcholine, palmitate, and TGFbeta, all of which have a role in liver injury, do not affect AnxA6 expression in human hepatocytes. Likewise, adiponectin and IL8 do not alter AnxA6 levels in primary human hepatic stellate cells. However, in hepatic tumors of 18 patients, AnxA6 protein levels are substantially reduced compared to nontumorous tissues. AnxA6 mRNA is even increased in the tumors suggesting that posttranscriptional mechanisms are involved herein. Lipidomic analysis shows trends toward elevated cholesteryl ester and sphingomyelin in the tumor samples, yet the ratio of tumor to nontumorous AnxA6 does not correlate with these lipids. The current study shows that AnxA6 is specifically reduced in human hepatocellular carcinoma suggesting a role of this protein in hepatocarcinogenesis.




Similar content being viewed by others
References
Buechler C, Wanninger J, Neumeier M (2011) Adiponectin, a key adipokine in obesity related liver diseases. World J Gastroenterol 17:2801–2811. doi:10.3748/wjg.v17.i23.2801
Clark JM (2006) The epidemiology of nonalcoholic fatty liver disease in adults. J Clin Gastroenterol 40(Suppl 1):S5–S10
Tagoe CE, Boustead CM, Higgins SJ, Walker JH (1994) Characterization and immunolocalization of rat liver annexin VI. Biochim Biophys Acta 1192:272–280
Enrich C, Rentero C, de Muga SV, Reverter M, Mulay V, Wood P, Koese M, Grewal T (2011) Annexin A6-linking Ca(2+) signaling with cholesterol transport. Biochim Biophys Acta 1813:935–947. doi:10.1016/j.bbamcr.2010.09.015
Grewal T, Koese M, Rentero C, Enrich C (2010) Annexin A6-regulator of the EGFR/Ras signalling pathway and cholesterol homeostasis. Int J Biochem Cell Biol 42:580–584. doi:10.1016/j.biocel.2009.12.020
Qi H, Liu S, Guo C, Wang J, Greenaway FT, Sun MZ (2015) Role of annexin A6 in cancer. Oncol Lett 10:1947–1952. doi:10.3892/ol.2015.3498
de Diego I, Schwartz F, Siegfried H, Dauterstedt P, Heeren J, Beisiegel U, Enrich C, Grewal T (2002) Cholesterol modulates the membrane binding and intracellular distribution of annexin 6. J Biol Chem 277:32187–32194. doi:10.1074/jbc.M205499200
Grewal T, Heeren J, Mewawala D, Schnitgerhans T, Wendt D, Salomon G, Enrich C, Beisiegel U, Jackle S (2000) Annexin VI stimulates endocytosis and is involved in the trafficking of low density lipoprotein to the prelysosomal compartment. J Biol Chem 275:33806–33813. doi:10.1074/jbc.M002662200
Pons M, Grewal T, Rius E, Schnitgerhans T, Jackle S, Enrich C (2001) Evidence for the Involvement of annexin 6 in the trafficking between the endocytic compartment and lysosomes. Exp Cell Res 269:13–22. doi:10.1006/excr.2001.5268
Hendrikx T, Walenbergh SM, Hofker MH, Shiri-Sverdlov R (2014) Lysosomal cholesterol accumulation: driver on the road to inflammation during atherosclerosis and non-alcoholic steatohepatitis. Obes Rev 15:424–433. doi:10.1111/obr.12159
Reverter M, Rentero C, de Muga SV, Alvarez-Guaita A, Mulay V, Cairns R, Wood P, Monastyrskaya K, Pol A, Tebar F, Blasi J, Grewal T, Enrich C (2011) Cholesterol transport from late endosomes to the Golgi regulates t-SNARE trafficking, assembly, and function. Mol Biol Cell 22:4108–4123. doi:10.1091/mbc.E11-04-0332
Garcia-Melero A, Reverter M, Hoque M, Meneses-Salas E, Koese M, Conway JR, Johnsen CH, Alvarez-Guaita A, Morales-Paytuvi F, Elmaghrabi YA, Pol A, Tebar F, Murray RZ, Timpson P, Enrich C, Grewal T, Rentero C (2015) Annexin A6 and late endosomal cholesterol modulates integrin recycling and cell migration. J Biol Chem. doi:10.1074/jbc.M115.683557
Sorrentino P, Terracciano L, D’Angelo S, Ferbo U, Bracigliano A, Vecchione R (2010) Predicting fibrosis worsening in obese patients with NASH through parenchymal fibronectin, HOMA-IR, and hypertension. Am J Gastroenterol 105:336–344. doi:10.1038/ajg.2009.587
Tilg H (2010) The role of cytokines in non-alcoholic fatty liver disease. Dig Dis 28:179–185. doi:10.1159/000282083
Grewal T, Enrich C (2009) Annexins–modulators of EGF receptor signalling and trafficking. Cell Signal 21:847–858
Berasain C, Avila MA (2014) The EGFR signalling system in the liver: from hepatoprotection to hepatocarcinogenesis. J Gastroenterol 49:9–23. doi:10.1007/s00535-013-0907-x
Koumangoye RB, Nangami GN, Thompson PD, Agboto VK, Ochieng J, Sakwe AM (2013) Reduced annexin A6 expression promotes the degradation of activated epidermal growth factor receptor and sensitizes invasive breast cancer cells to EGFR-targeted tyrosine kinase inhibitors. Mol Cancer 12:167. doi:10.1186/1476-4598-12-167
Koese M, Rentero C, Kota BP, Hoque M, Cairns R, Wood P, Vila de Muga S, Reverter M, Alvarez-Guaita A, Monastyrskaya K, Hughes WE, Swarbrick A, Tebar F, Daly RJ, Enrich C, Grewal T (2013) Annexin A6 is a scaffold for PKCalpha to promote EGFR inactivation. Oncogene 32:2858–2872. doi:10.1038/onc.2012.303
Vila de Muga S, Timpson P, Cubells L, Evans R, Hayes TE, Rentero C, Hegemann A, Reverter M, Leschner J, Pol A, Tebar F, Daly RJ, Enrich C, Grewal T (2009) Annexin A6 inhibits Ras signalling in breast cancer cells. Oncogene 28:363–377. doi:10.1038/onc.2008.386
Stogbauer F, Weigert J, Neumeier M, Wanninger J, Sporrer D, Weber M, Schaffler A, Enrich C, Wood P, Grewal T, Aslanidis C, Buechler C (2009) Annexin A6 is highly abundant in monocytes of obese and type 2 diabetic individuals and is downregulated by adiponectin in vitro. Exp Mol Med 41:501–507. doi:10.3858/emm.2009.41.7.055
Arita Y, Kihara S, Ouchi N, Takahashi M, Maeda K, Miyagawa J, Hotta K, Shimomura I, Nakamura T, Miyaoka K, Kuriyama H, Nishida M, Yamashita S, Okubo K, Matsubara K, Muraguchi M, Ohmoto Y, Funahashi T, Matsuzawa Y (2012) Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. 1999. Biochem Biophys Res Commun 425:560–564. doi:10.1016/j.bbrc.2012.08.024
Wieser V, Moschen AR, Tilg H (2012) Adipocytokines and hepatocellular carcinoma. Dig Dis 30:508–513. doi:10.1159/000341702
Jia X, Yin L, Feng Y, Peng X, Ma F, Yao Y, Liu X, Zhang Z, Yuan Z, Zhang L (2012) A dynamic plasma membrane proteome analysis of alcohol-induced liver cirrhosis. Proteome Sci 10:39. doi:10.1186/1477-5956-10-39
Weigert J, Neumeier M, Bauer S, Mages W, Schnitzbauer AA, Obed A, Groschl B, Hartmann A, Schaffler A, Aslanidis C, Scholmerich J, Buechler C (2008) Small-interference RNA-mediated knock-down of aldehyde oxidase 1 in 3T3-L1 cells impairs adipogenesis and adiponectin release. FEBS Lett 582:2965–2972. doi:10.1016/j.febslet.2008.07.034
Damm G, Pfeiffer E, Burkhardt B, Vermehren J, Nussler AK, Weiss TS (2013) Human parenchymal and non-parenchymal liver cell isolation, culture and characterization. Hepatol Int 7:951–958. doi:10.1007/s12072-013-9475-7
Krautbauer S, Eisinger K, Hader Y, Buechler C (2014) Free fatty acids and IL-6 induce adipocyte galectin-3 which is increased in white and brown adipose tissues of obese mice. Cytokine 69:263–271. doi:10.1016/j.cyto.2014.06.016
Krautbauer S, Eisinger K, Lupke M, Wanninger J, Ruemmele P, Hader Y, Weiss TS, Buechler C (2013) Manganese superoxide dismutase is reduced in the liver of male but not female humans and rodents with non-alcoholic fatty liver disease. Exp Mol Pathol 95:330–335. doi:10.1016/j.yexmp.2013.10.003
Krautbauer S, Eisinger K, Neumeier M, Hader Y, Buettner R, Schmid PM, Aslanidis C, Buechler C (2014) Free fatty acids, lipopolysaccharide and IL-1alpha induce adipocyte manganese superoxide dismutase which is increased in visceral adipose tissues of obese rodents. PLoS One 9:e86866. doi:10.1371/journal.pone.0086866
Krautbauer S, Wanninger J, Eisinger K, Hader Y, Beck M, Kopp A, Schmid A, Weiss TS, Dorn C, Buechler C (2013) Chemerin is highly expressed in hepatocytes and is induced in non-alcoholic steatohepatitis liver. Exp Mol Pathol 95:199–205. doi:10.1016/j.yexmp.2013.07.009
Park CW, Kim HW, Ko SH, Lim JH, Ryu GR, Chung HW, Han SW, Shin SJ, Bang BK, Breyer MD, Chang YS (2007) Long-term treatment of glucagon-like peptide-1 analog exendin-4 ameliorates diabetic nephropathy through improving metabolic anomalies in db/db mice. J Am Soc Nephrol 18:1227–1238. doi:10.1681/ASN.2006070778
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Liebisch G, Lieser B, Rathenberg J, Drobnik W, Schmitz G (2004) High-throughput quantification of phosphatidylcholine and sphingomyelin by electrospray ionization tandem mass spectrometry coupled with isotope correction algorithm. Biochim Biophys Acta 1686:108–117. doi:10.1016/j.bbalip.2004.09.003
Liebisch G, Binder M, Schifferer R, Langmann T, Schulz B, Schmitz G (2006) High throughput quantification of cholesterol and cholesteryl ester by electrospray ionization tandem mass spectrometry (ESI-MS/MS). Biochim Biophys Acta 1761:121–128. doi:10.1016/j.bbalip.2005.12.007
Liebisch G, Vizcaino JA, Kofeler H, Trotzmuller M, Griffiths WJ, Schmitz G, Spener F, Wakelam MJ (2013) Shorthand notation for lipid structures derived from mass spectrometry. J Lipid Res 54:1523–1530. doi:10.1194/jlr.M033506
Langhans B, Kramer B, Louis M, Nischalke HD, Huneburg R, Staratschek-Jox A, Odenthal M, Manekeller S, Schepke M, Kalff J, Fischer HP, Schultze JL, Spengler U (2013) Intrahepatic IL-8 producing Foxp3(+)CD4(+) regulatory T cells and fibrogenesis in chronic hepatitis C. J Hepatol 59:229–235. doi:10.1016/j.jhep.2013.04.011
Kakisaka K, Cazanave SC, Fingas CD, Guicciardi ME, Bronk SF, Werneburg NW, Mott JL, Gores GJ (2012) Mechanisms of lysophosphatidylcholine-induced hepatocyte lipoapoptosis. Am J Physiol Gastrointest Liver Physiol 302:G77–G84. doi:10.1152/ajpgi.00301.2011
Kwan HY, Fong WF, Yang Z, Yu ZL, Hsiao WL (2013) Inhibition of DNA-dependent protein kinase reduced palmitate and oleate-induced lipid accumulation in HepG2 cells. Eur J Nutr 52:1621–1630. doi:10.1007/s00394-012-0467-5
Eisinger K, Krautbauer S, Hebel T, Schmitz G, Aslanidis C, Liebisch G, Buechler C (2014) Lipidomic analysis of the liver from high-fat diet induced obese mice identifies changes in multiple lipid classes. Exp Mol Pathol 97:37–43. doi:10.1016/j.yexmp.2014.05.002
Eisinger K, Liebisch G, Schmitz G, Aslanidis C, Krautbauer S, Buechler C (2014) Lipidomic analysis of serum from high fat diet induced obese mice. Int J Mol Sci 15:2991–3002. doi:10.3390/ijms15022991
Schattenberg JM, Galle PR (2010) Animal models of non-alcoholic steatohepatitis: of mice and man. Dig Dis 28:247–254. doi:10.1159/000282097
Wanninger J, Walter R, Bauer S, Eisinger K, Schaffler A, Dorn C, Weiss TS, Hellerbrand C, Buechler C (2011) MMP-9 activity is increased by adiponectin in primary human hepatocytes but even negatively correlates with serum adiponectin in a rodent model of non-alcoholic steatohepatitis. Exp Mol Pathol 91:603–607. doi:10.1016/j.yexmp.2011.07.001
Slotte JP (2013) Biological functions of sphingomyelins. Prog Lipid Res 52:424–437. doi:10.1016/j.plipres.2013.05.001
Lee JS, Heo J, Libbrecht L, Chu IS, Kaposi-Novak P, Calvisi DF, Mikaelyan A, Roberts LR, Demetris AJ, Sun Z, Nevens F, Roskams T, Thorgeirsson SS (2006) A novel prognostic subtype of human hepatocellular carcinoma derived from hepatic progenitor cells. Nat Med 12:410–416. doi:10.1038/nm1377
Lee YJ, Hah YJ, Kang YN, Kang KJ, Hwang JS, Chung WJ, Cho KB, Park KS, Kim ES, Seo HY, Kim MK, Park KG, Jang BK (2013) The autophagy-related marker LC3 can predict prognosis in human hepatocellular carcinoma. PLoS One 8:e81540. doi:10.1371/journal.pone.0081540
Wang X, Zhang S, Zhang J, Lam E, Liu X, Sun J, Feng L, Lu H, Yu J, Jin H (2013) Annexin A6 is down-regulated through promoter methylation in gastric cancer. Am J Transl Res 5:555–562
Qi Y, Zhang X, Kang Y, Wu J, Chen J, Li H, Guo Y, Liu B, Shao Z, Zhao X (2015) Genome-wide transcriptional profiling analysis reveals annexin A6 as a novel EZH2 target gene involving gastric cellular proliferation. Mol BioSyst 11:1980–1986. doi:10.1039/c5mb00233h
Della Gaspera B, Braut-Boucher F, Bomsel M, Chatelet F, Guguen-Guillouzo C, Font J, Weinman J, Weinman S (2001) Annexin expressions are temporally and spatially regulated during rat hepatocyte differentiation. Dev Dyn 222:206–217. doi:10.1002/dvdy.1183
Marengo A, Rosso C, Bugianesi E (2015) Liver Cancer: connections with Obesity, Fatty Liver, and Cirrhosis. Annu Rev Med. doi:10.1146/annurev-med-090514-013832
Eggens I, Ekstrom TJ, Aberg F (1990) Studies on the biosynthesis of polyisoprenols, cholesterol and ubiquinone in highly differentiated human hepatomas. J Exp Pathol (Oxford) 71:219–232
Lu M, Hu XH, Li Q, Xiong Y, Hu GJ, Xu JJ, Zhao XN, Wei XX, Chang CC, Liu YK, Nan FJ, Li J, Chang TY, Song BL, Li BL (2013) A specific cholesterol metabolic pathway is established in a subset of HCCs for tumor growth. J Mol Cell Biol 5:404–415. doi:10.1093/jmcb/mjt039
Cubells L, Vila de Muga S, Tebar F, Wood P, Evans R, Ingelmo-Torres M, Calvo M, Gaus K, Pol A, Grewal T, Enrich C (2007) Annexin A6-induced alterations in cholesterol transport and caveolin export from the Golgi complex. Traffic 8:1568–1589. doi:10.1111/j.1600-0854.2007.00640.x
Ridgway ND (2000) Interactions between metabolism and intracellular distribution of cholesterol and sphingomyelin. Biochim Biophys Acta 1484:129–141
Acknowledgments
C. B. is supported by the German Research Foundation (BU1141/7-1 and BU1141/8-1) and the “Stiftung für Pathobiochemie und Molekulare Diagnostik.” A.J.H is supported by a Helen and Robert Ellis Postdoctoral Research Fellowship from the Sydney Medical School Foundation and funding from the University of Sydney. T.G acknowledges the support of the University of Sydney, Australia (U7007, U7042). The technical assistance of Yvonne Hader, Jolante Aiwanger, Simone Düchtel, and Doreen Müller is greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Meier, E.M., Rein-Fischboeck, L., Pohl, R. et al. Annexin A6 protein is downregulated in human hepatocellular carcinoma. Mol Cell Biochem 418, 81–90 (2016). https://doi.org/10.1007/s11010-016-2735-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-016-2735-9