Abstract
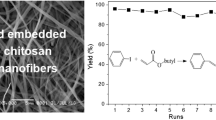
In this paper, chitosan composite nanofibers with mean diameters of 300–500 nm have been prepared by electrospinning with poly(methacrylic acid) as the crosslinking agent and poly(ethylene oxide) as the cospinning polymer. Afterwards, these composite nanofibers were annealed at temperatures ranging from 110 to 190 °C to improve their solvent resistance. Their morphologies and structures were characterized by scanning electron microscopy, infrared spectroscopy (IR) and positron annihilation life spectroscopy. These composite nanofibers were used to adsorb the Pd2+ cations in aqueous solutions. The effects of chitosan content, annealing temperature, adsorption temperature, solution pH and initial Pd2+ concentration were carefully studied. The adsorption result shows that these composite nanofibers had better adsorption performance than the pristine chitosan and commercial activated carbon. Under the optimized adsorption conditions, the maximum adsorption capacity was found to be 366 mg/g. The adsorption kinetic, FT-IR and XPS confirmed the chemical adsorption behavior of Pd2+ cations on the composite nanofibers. The thermodynamic parameters (ΔG0, ΔH0 and ΔS0) acquired by Van’t Hoff equation indicated that the adsorption process was spontaneous and exothermic in nature. Moreover, selective adsorption study revealed that these composite nanofibers exhibited good adsorption selectivity for Pd2+ cations from the mixture of metal ions in aqueous solution. At last, the reusability of these novel composite nanofibers was tested for four runs. Overall, this study indicated that the as-prepared nanofiber mat was an efficient adsorbent for the recovery of precious palladium cations from aqueous solutions.
















Similar content being viewed by others
References
Abiman P, Wildgoose GG, Crossley A, Compton RG (2008) Removal of palladium ions from aqueous systems by chemically modified cysteine carbon powder. J Mater Chem 18:3948–3953. https://doi.org/10.1039/B805804K
Alves DCDS, Healy B, Pinto LADA, Cadaval TRS Jr, Breslin CB (2021) Recent developments in chitosan-based adsorbents for the removal of pollutants from aqueous environments. Molecules 26:594. https://doi.org/10.3390/molecules26030594
Asere TG, Mincke S, Folens K, Bussche FV, Lapeire L, Verbeken K, Voort PVD, Tessema DA, Laing GD, Stevens C (2019) Dialdehyde carboxymethyl cellulose cross-linked chitosan for the recovery of palladium and platinum from aqueous solution. React Funct Polym 141:145–154. https://doi.org/10.1016/j.reactfunctpolym.2019.05.008
Chang SH (2020) Gold(III) recovery from aqueous solutions by raw and modified chitosan: a review. Carbohydr Polym 256:117423. https://doi.org/10.1016/j.carbpol.2020.117423
Das N (2010) Recovery of precious metals through biosorption—a review. Hydrometallurgy 103:180–189. https://doi.org/10.1016/j.hydromet.2010.03.016
Ei-Aassar MR, Mohamed FM, Alsohaimi IH, Khalifa RE (2021) Fabrication of novel valorized ecofriendly olive seed residue/ anthracite/chitosan composite for removal of Cr (VI): kinetics, isotherms and thermodynamics modeling. Cellulose 28:7165–7183. https://doi.org/10.1007/s10570-021-03963-y
El-Gamal S, Elsayed M (2020) Synthesis, structural, thermal, mechanical, and nano-scale free volume properties of novel PbO/PVC/PMMA nanocomposites. Polymer 206:122911. https://doi.org/10.1016/j.polymer.2020.122911
Fraser JG, Beamish FE, Mcbryde W (1954) Separation of palladium from lead and colorimetric determination of palladium with potassium iodide. Anal Chem 26:495–498. https://doi.org/10.1021/ac60087a020
Fujiwara K, Ramesh A, Maki T, Hasegawa H, Ueda K (2017) Adsorption of platinum (IV), palladium (II) and gold (III) from aqueous solutions onto L-lysine modified crosslinked chitosan resin. J Hazard Mater 146:39–50. https://doi.org/10.1016/j.jhazmat.2006.11.049
Gandhi MR, Yamada M, Haga K, Shibayama A (2017) Synthesis of pincer-type extractants for selective extraction of palladium from PGMs: an improved liquid-liquid extraction approach to current refining processes. Sci Rep 7:8709. https://doi.org/10.1038/s41598-017-09053-z
Habiba U, Siddique TA, Lee JL, Joo TC, Ang BC, Afifi AM (2018) Adsorption study of methyl orange by chitosan/polyvinyl alcohol/zeolite electrospun composite nanofibrous membrane. Carbohydr Polym 191:79–85. https://doi.org/10.1016/j.carbpol.2018.02.081
Ito Y, Mohamed HFM, Shiotani M (1996) Vacancies in the solids of low molecular weight organic compounds observed by positron annihilation. J Phys Chem 100:14161–14165. https://doi.org/10.1021/jp9604002
Jiang M, Han T, Wang J, Shao L, Qi C, Zhang XM, Liu C, Liu X (2018) Removal of heavy metal chromium using cross-linked chitosan composite nanofiber mats. Int J Biol Macromol 120:213–221. https://doi.org/10.1016/j.ijbiomac.2018.08.071
Katsutoshi I, Manju G, Ying X, Kawakita H, Ohto K, Alam S (2015) Hydrometallurgical recovery of precious metals and removal of hazardous metals using persimmon tannin and persimmon wastes. Metals 5:1921–1956. https://doi.org/10.3390/met5041921
Li L, Zhang J, Li Y, Yang C (2017) Removal of Cr (VI) with a spiral wound chitosan nanofiber membrane module via dead-end filtration. J Membr Sci 544:333–341. https://doi.org/10.1016/j.memsci.2017.09.045
Lin S, Wei W, Wu X, Zhou T, Mao J, Yun YS (2015) Selective recovery of Pd(II) from extremely acidic solution using ion-imprinted chitosan fiber: adsorption performance and mechanisms. J Hazard Mater 299:10–17. https://doi.org/10.1016/j.jhazmat.2015.05.050
Mansor N, Mohamed N, Suah FBM (2021) Electrochemical recovery of low concentration of palladium from palladium(II) chloride of electroplating wastewater. J Chem Technol Biot 96:3216–3223. https://doi.org/10.1002/jctb.6878
Militello MC (1994) Palladium chloride (PdCl2) by XPS. Surf Sci Spectra 3:402–409. https://doi.org/10.1116/1.1247785
Monier M, Abdel-Latif DA (2017) Fabrication of Au(III) ion-imprinted polymer based on thiol-modified chitosan. Int J Biol Macromol 105:777–787. https://doi.org/10.1016/j.ijbiomac.2017.07.098
Nagireddi S, Katiyar V, Uppaluri R (2017) Pd(II) adsorption characteristics of glutaraldehyde cross-linked chitosan copolymer resin. Int J Biol Macromol 94:72–84. https://doi.org/10.1016/j.ijbiomac.2016.09.088
Nuruddin M, Chowdhury RA, Lopez-Perez N, Montes FJ, Youngblood JP, Howarter JA (2020) Influence of free volume determined by positron annihilation lifetime spectroscopy (PALS) on gas permeability of cellulose nanocrystal films. ACS Appl Mater Interfaces 12:24380–24389. https://doi.org/10.1021/acsami.0c05738
Omrani M, Goriaux M, Liu Y, Martinet S, Jean-Soro L, Ruban V (2020) Platinum group elements study in automobile catalysts and exhaust gas samples. Environ Pollut 257:113477. https://doi.org/10.1016/j.envpol.2019.113477
Petrova YS, Pestov AV, Usoltseva MK, Kapitanova E, Neudachina LK (2019) Methods for correction of selectivity of N -(2-sulfoethyl)chitosan-based materials towards platinum(IV) and palladium(II) ions. Sep Sci Technol 54:1–9. https://doi.org/10.1080/01496395.2018.1505912
Ruiz M, Sastre AM, Guibal E (2000) Palladium sorption on glutaraldehyde-crosslinked chitosan. React Funct Polym 45:155–173. https://doi.org/10.1016/S1381-5148(00)00019-5
Shao L, Ren Y, Wang Z, Qi C, Lin Y (2015) Developing chitosan-based composite nanofibers for supporting metal catalysts. Polymer 75:168–177. https://doi.org/10.1016/j.polymer.2015.08.031
Sharma S, Kumar AK, Rajesh N (2017) A perspective on diverse adsorbent materials to recover precious palladium and the way forward. RSC Adv 7:52133–52142. https://doi.org/10.1039/C7RA10153H
Shen N, Chirwa E (2020) Live and lyophilized fungi-algae pellets as novel biosorbents for gold recovery: critical parameters, isotherm, kinetics and regeneration studies. Bioresour Technol 306:123041. https://doi.org/10.1016/j.biortech.2020.123041
Silva AO, Cunha RS, Hotza D, Machado RAF (2021) Chitosan as a matrix of nanocomposites: a review on nanostructures, processes, properties, and applications. Carbohydr Polym 272:118472. https://doi.org/10.1016/j.carbpol.2021.118472
Tao SJ (1972) Positronium annihilation in molecular substances. J Chem Phys 56:5499–5510. https://doi.org/10.1063/1.1677067
Tofan L, Wenkert R (2020) Chelating polymers with valuable sorption potential for development of precious metal recycling technologies. Rev Chem Eng. https://doi.org/10.1515/revce-2019-0075.10.1515/revce-2019-0075
Veerakumar P, Lin KC (2020) An overview of palladium supported on carbon-based materials: synthesis, characterization, and its catalytic activity for reduction of hexavalent chromium. Chemosphere 253:126750. https://doi.org/10.1016/j.chemosphere.2020.126750
Viltres H, López YC, Levya C, Gupta NK, Naranjo AG, Acevedo-Peña S-D, Bae J, Kim KS (2021) Polyamidoamine dendrimer-based materials for environmental applications: a review. J Mol Liq 334:116017. https://doi.org/10.1016/j.molliq.2021.116017
Wu D, Lu D, Sun F, Zhou Y (2019) Process optimization for simultaneous antibiotic removal and precious metal recovery in an energy neutral process. Sci Total Environ 695:133914.1-133914.8. https://doi.org/10.1016/j.scitotenv.2019.133914
Xing G, Shao L, Du Y, Qi C (2021) Citric acid crosslinked chitosan/poly(ethylene oxide) composite nanofibers fabricated by electrospinning and thermal treatment for controlled drug release. Cellulose 28:961–971. https://doi.org/10.1007/s10570-020-03562-3
Zhou Z, Liu F, Huang Y, Wang Z, Li G (2015) Biosorption of palladium(II) from aqueous solution by grafting chitosan on persimmon tannin extract. Int J Biol Macromol 77:336–343. https://doi.org/10.1016/j.ijbiomac.2015.03.037
Acknowledgments
The authors acknowledge the financial support from the National Natural Science Foundation of China (Nos. 12075154 and 11905132).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, Q., Luo, Y., Wang, D. et al. Fabrication of chitosan composite nanofibers for the recovery of precious palladium cations from aqueous solution. Cellulose 29, 5803–5816 (2022). https://doi.org/10.1007/s10570-022-04628-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-022-04628-0