Abstract
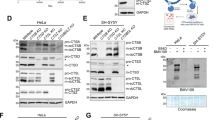
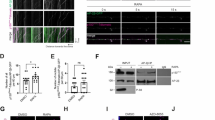
Store-operated calcium entry (SOCE) channels composed of Stim and Orai proteins play a critical role in diverse biological processes. Upon endoplasmic reticulum (ER)-mediated calcium (Ca2+) depletion, Stim proteins oligomerize with Orai to initiate Ca2+ influx across the plasma membrane. The ubiquitin-like (UBL) and ubiquitin-associated (UBA) domains of ubiquilin 1 are involved in the degradation of presenilin and polyglutamine proteins. Through screening of Orai1 interaction partner(s) that might have an effect on SOCE, ubiquilin 1 was identified as a target of Orai1. However, the UBL and UBA domains of ubiquilin 1 were dispensable for this interaction. Additionally, ubiquilin 1 and Orai1 colocalized in the cytosolic compartment. Ubiquilin 1 increased the ubiquitination of Orai1, resulting in the formation of a high-molecular-weight form. MG132, a proteasome inhibitor, failed to block the degradation of Orai1, whereas bafilomycin A, a lysosome inhibitor, prevented Orai1 degradation. Confocal microscopy studies demonstrated that a fraction of Orai1 colocalized with ubiquilin 1 and the autophagosomal marker LC3. Because Orai1 is a constituent of SOCE, we determined the effect of ubiquilin 1 on Orai1-mediated Ca2+ influx. As we expected, intracellular Ca2+ mobilization, a process normally potentiated by Orai1, was downregulated by ubiquilin 1. Taken together, these findings suggest that ubiquilin 1 downregulates intracellular Ca2+ mobilization and its downstream signaling by promoting the ubiquitination and lysosomal degradation of Orai1.
Similar content being viewed by others
References
Augustine, G.J., Santamaria, F., and Tanaka, K. (2003). Local calcium signaling in neurons. Neuron 40, 331–346.
Berridge, M.J., Lipp, P., and Bootman, M.D. (2000). Signal transduction. The calcium entry pas de deux. Science 287, 1604–1605.
Biswas, N., Liu, S., Ronni, T., Aussenberg, S.E., Liu, W., Fujita, T., and Wang, T. (2011). The ubiquitin-like protein PLIC-1 or ubiquilin 1 inhibits TLR3-Trif signaling. PLoS One 6, e21153.
Clapham, D.E. (2007). Calcium signaling. Cell 131, 1047–1058.
Decuypere, J.P., Bultynck, G., and Parys, J.B. (2011). A dual role for Ca2+ in autophagy regulation. Cell Calcium 50, 242–250.
El, A.A., Stieren, E.S., Barral, J.M., and Boehning, D. (2012). Ubiquilin-1 regulates amyloid precursor protein maturation and degradation by stimulating K63-linked polyubiquitination of lysine 688. Proc. Natl. Acad. Sci. USA 109, 13416–13421.
Eylenstein, A., Gehring, E.M., Heise, N., Shumilina, E., Schmidt, S., Szteyn, K., Munzer, P., Nurbaeva, M.K., Eichenmuller, M., Tyan, L., et al. (2011). Stimulation of Ca2+-channel Orai1/STIM1 by serum- and glucocorticoid-inducible kinase 1 (SGK1). FASEB J. 25, 2012–2021.
Ficklin, M.B., Zhao, S.l., and Feng, G.P. (2005). Ubiquilin-1 regulates nicotine-induced up-regulation of neuronal nicotinic acetylcholine receptors. J. Biol. Chem. 280, 34088–34095.
Flourakis, M., Lehen’kyi, V., Beck, B., Raphael, M., Vandenberghe, M., Abeele, F.V., Roudbaraki, M., Lepage, G., Mauroy, B., Romanin, C., et al. (2010). Orai1 contributes to the establishment of an apoptosis-resistant phenotype in prostate cancer cells. Cell Death Dis. 1, e75.
Hogan, P.G., Lewis, R.S., and Rao, A. (2010). Molecular basis of calcium signaling in lymphocytes: STIM and ORAI. Annu. Rev. Immunol. 28, 491–533.
Jeremy, T.S., Amber, M.B, Shilan, W., James, W.P., and Nasser, M.R. (2012). Phosphoregulation of STIM1 leads to exclusion of the endoplasmic reticulum from the mitotic spindle. Curr. Biol. 22, 1487–1493.
Kawasaki, T., Ueyama, T., Lange, I., Feske, S., and Saito, N. (2010). Protein kinase C-induced phosphorylation of Orai1 regulates the intracellular Ca2+ level via the store-operated Ca2+ channel. J. Biol. Chem. 285, 25720–25730.
Kim, H.R., Oh, B.C., Choi, J.K., and Bae, S.C. (2008). Pim-1 kinase phosphorylates and stabilizes RUNX3 and alters its subcellular localization. J. Cell Biochem. 105, 1048–1058.
Krapivinsky, G., Krapivinsky, L., Stotz, S.C., Manasian, Y., and Clapham, D.E. (2011). POST, partner of stromal interaction molecule 1 (STIM1), targets STIM1 to multiple transporters. Proc. Natl. Acad. Sci. USA 108, 19234–19239.
Kurosaki, T., and Baba, Y. (2010). Ca2+ signaling and STIM1. Prog. Biophys. Mol. Biol. 103, 51–58.
Lee, K.Y., Lee, J.W., Nam, H.J., Shim, J.H., Song, Y.S., and Kang, K.W., (2011). PI3-kinase/p38 kinase-dependent E2F1 activation is critical for Pin1 induction in Tamoxifen-resistant breast cancer cells. Mol. Cells 32, 107–111.
Lee, H.J., Bae, G.U., Leem, Y.E., Choi, H.K., Kang, T.M., Cho, H., Kim, S.T., and Kang, J.S. (2012). Phosphorylation of Stim1 at serine 575 via netrin-2/Cdo-activated ERK1/2 is critical for the promyogenic function of Stim1. Mol. Biol. Cell 23, 1376–1387.
Limnander, A., Depeille, P., Freedman, T.S., Liou, J., Leitges, M., Kurosaki, T., Roose, J.P., and Weiss, A. (2011). STIM1, PKCdelta and RasGRP set a threshold for proapoptotic Erk signaling during B cell development. Nat. Immunol. 12, 425–433.
Liou, J., Kim, M.L., Heo, W.D., Jones, J.T., Myers, J.W., Ferrell, J.E. Jr., and Meyer, T. (2005). STIM is a Ca2+ sensor essential for Ca2+-store-depletion-triggered Ca2+ influx. Curr. Biol. 15, 1235–1241.
Pozo, G.E., Campbell, D.G., Deak, M., varez-Barrientos, A., Morrice, N.A., Alvarez, I.S., Alessi, D.R., and Martin-Romero, F.J. (2010). Phosphorylation of STIM1 at ERK1/2 target sites modulates store-operated calcium entry. J. Cell Sci. 123, 3084–3093.
Rothenberg, C., Srinivasan, D., Mah, L., Kaushik. S., Peterhoff, C. M., Ugolino, J., Fang, S., Maria, C.A., Nixon, R.A., and Monteiro, M.J. (2010). Ubiquilin functions in autophagy and is degraded by chaperone-mediated autophagy. Hum. Mol. Genet. 19, 3219–3232.
Sathish, V., Abcejo, A.J., Thompson, M.A., Sieck, G.C., Prakash, Y.S., and Pabelick, C.M. (2012). Caveolin-1 regulation of storeoperated Ca2+ influx in human airway smooth muscle. Eur. Respir. J. 40, 470–478.
Schubert, T., Weiler, R., and Feigenspan, A. (2006). Intracellular calcium is regulated by different pathways in horizontal cells of the mouse retina. J. Neurophysiol. 96, 1278–1292.
Srikanth, S., Jew, M., Kim, K.D., Yee, M.K., Abramson, J., and Gwack, Y. (2012). Junctate is a Ca2+-sensing structural component of Orai1 and stromal interaction molecule 1 (STIM1). Proc. Natl. Acad. Sci. USA 109, 8682–8687.
Varnai, P., Hunyady, L., and Balla, T. (2009). STIM and Orai: the long-awaited constituents of store-operated calcium entry. Trends Pharmacol. Sci. 30, 118–128.
Viswanathan, J., Haapasalo, A., Bottcher, C., Miettinen, R., Kurkinen, K.M., Lu, A., Thomas, A., Maynard, C.J., Romano, D., Hyman, B.T., et al. (2011). Alzheimer’s disease-associated ubiquilin-1 regulates presenilin-1 accumulation and aggresome formation. Traffic 12, 330–348.
Walsh, C.M., Doherty, M.K., Tepikin, A.V., and Burgoyne, R.D. (2010). Evidence for an interaction between Golli and STIM1 in store-operated calcium entry. Biochem. J. 430, 453–460.
Wang, H., and Monteiro, M.J. (2007). Ubiquilin overexpression reduces GFP-polyalanine-induced protein aggregates and toxicity. Exp. Cell Res. 313, 2810–2820.
Wu, M.M., Luik, R.M., Lewis, R.S., (2007). Some assembly required: constructing the elementary units of store-operated Ca2+ entry. Cell Calcium 42, 163–172.
Zhang, S.L., Yu, Y., Roos, J., Kozak, J.A., Deerinck, T.J., Ellisman, M.H., Stauderman, K.A., and Cahalan, M.D. (2005). STIM1 is a Ca2+ sensor that activates CRAC channels and migrates from the Ca2+ store to the plasma membrane. Nature 437, 902–905.
Zhou, Y.B., Ramachandran, S., Oh-Hora, M., Rao, A., and Hogan, P.G. (2010). Pore architecture of the ORAI1 store-operated calcium channel. Proc. Natl. Acad. Sci. USA 107, 4896–4901.
Author information
Authors and Affiliations
Corresponding authors
About this article
Cite this article
Lee, JE., Jeon, IS., Han, NE. et al. Ubiquilin 1 interacts with Orai1 to regulate calcium mobilization. Mol Cells 35, 41–46 (2013). https://doi.org/10.1007/s10059-013-2268-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-013-2268-7