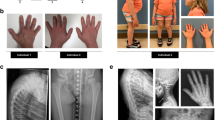
Abstract
The secreted polypeptide noggin (encoded by the Nog gene) binds and inactivates members of the transforming growth factor β superfamily of signalling proteins (TGFβ-FMs), such as BMP4 (ref. 1). By diffusing through extracellular matrices more efficiently than TGFβ-FMs, noggin may have a principal role in creating morphogenic gradients2. During mouse embryogenesis, Nog is expressed at multiple sites3, including developing bones4,5. Nog-/- mice die at birth from multiple defects that include bony fusion of the appendicular skeleton3,4. We have identified five dominant human NOG mutations in unrelated families segregating proximal symphalangism (SYM1; OMIM 185800) and a de novo mutation in a patient with unaffected parents. We also found a dominant NOG mutation in a family segregating multiple synostoses syndrome (SYNS1; OMIM 186500); both SYM1 and SYNS1 have multiple joint fusion as their principal feature6,7. All seven NOG mutations alter evolutionarily conserved amino acid residues. The findings reported here confirm that NOG is essential for joint formation and suggest that NOG requirements during skeletogenesis differ between species and between specific skeletal elements within species.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Zimmerman, L.B., De Jesus-Escobar, J.M. & Harland, R.M. The Spemann organizer signal noggin binds and inactivates bone morphogenetic protein 4. Cell 86, 599–606 (1996).
Jones, C.M. & Smith, J.C. Establishment of a BMP-4 morphogen gradient by long-range inhibition. Dev. Biol. 194, 12–17 (1998).
McMahon, J.A. et al. Noggin-mediated antagonism of BMP signaling is required for growth and patterning of the neural tube and somite. Genes Dev. 12, 1438–1452 (1998).
Brunet, L.J., McMahon, J.A., McMahon, A.P. & Harland, R.M. Noggin, cartilage morphogenesis, and joint formation in the mammalian skeleton. Science 280, 1455–1457 (1998).
Capdevila, J. & Johnson R.L. Endogenous and ectopic expression of noggin suggests a conserved mechanism for regulation of BMP function during limb and somite patterning. Dev. Biol. 197, 205–217 (1998).
Polymeropoulos, M.H., Poush, J., Rubenstein, J.R. & Francomano, C.A. Localization of the gene (SYM1) for proximal symphalangism to human chromosome 17q21-q22. Genomics 27, 225– 229 (1995).
Krakow, D. et al. Localization of a multiple synostoses-syndrome disease gene to chromosome 17q21-22. Am. J. Hum. Genet. 63, 120–124 (1998).
Polinkovsky, A. et al. Mutations in CDMP1 cause autosomal dominant brachydactyly type C. Nature Genet. 17, 18– 19 (1997).
Valenzuela, D.M. et al. Identification of mammalian noggin and its expression in the adult nervous system. J. Neurosci. 15, 6077 –6084 (1995).
Strasburger, A.K., Hawkins, M.R., Eldridge, R., Hargrave, R.L. & McKusick, V.A. Symphalangism: genetic and clinical aspects. Bull. Johns Hopkins Hosp. 117, 108–127 (1965).
Park, J.P., Moeschler, J.B., Berg, S.Z., Bauer, R.M. & Wurster-Hill, D.H. A unique de novo interstitial deletion del(17)(q21.3q23) in a phenotypically abnormal infant. Clin. Genet. 41, 54–56 (1992).
Dallapiccola, B., Mingarelli, R., Digilio, C., Obregon, M.G. & Giannotti, A. Interstitial deletion del(17)(q21.3q23 or 24.2) syndrome. Clin. Genet. 43, 54– 55 (1993).
Khalifa, M.M., MacLeod, P.M. & Duncan, A.M.V. Additional case of de novo interstitial deletion del(17)(q21.3q23) and expansion of the phenotype. Clin. Genet. 44, 258–261 (1993).
Smith, W.C., Knecht, A.K., Wu, M. & Harland, R.M. Secreted noggin protein mimics the Spemann organizer in dorsalizing Xenopus mesoderm. Nature 361, 547– 549 (1993).
Holley, S.A. et al. The Xenopus dorsalizing factor noggin ventralizes drosophila embryos by preventing DPP from activating its receptor. Cell 86, 607–617 (1996).
Nakamura, R. et al. Activin-binding protein from rat ovary is follistatin. Science 247, 836–838 (1990).
Piccolo, S., Yoshiki, S., Lu, B. & De Robertis, E.M. Dorsoventral patterning in Xenopus: inhibition of ventral signals by direct binding of chordin to BMP-4. Cell 86, 589– 598 (1996).
Fainsod, A. et al. The dorsalizing and neural inducing gene follistatin is an antagonist of BMP-4. Mech. Dev. 63, 39–50 (1997).
Thomas, J.T. et al. Disruption of limb morphogenesis by a dominant negative mutation in CDMP1. Nature Genet. 17, 58– 64 (1997).
Storm, E.E., et al. Limb alterations in brachypodism mice due to mutations in a new member of the TGF b-superfamily. Nature, 368, 693–643 (1994).
Witta, S.E., Agarwal, V.R. & Sato, S.M. XlPOU2, a noggin-inducible gene, has direct neuralizing activity. Development 121, 721– 730 (1995).
de Kok, Y.J.M. et al. Association between X-linked mixed deafness and mutations in the POU domain gene POU3F4. Science 267, 685–688 (1995).
Ensink, R.J.H., Sleeckx, J.P. & Cremers, C.W.R.J. Proximal symphalangism and congenital conductive hearing loss; otological aspects. Am. J. Otol. (in press).
Cremers, C., Theunissen, E. & Kuijpers, W. Proximal symphalangism and stapes ankylosis. Arch. Otolaryngol. 111, 765–767 (1985).
Acknowledgements
We thank the patients and their families for participating; V. McKusick and M. Abbott for providing Fig. 1b; and J. Hurvitz, R.T. Ballock, E. Eichler, I. McIntosh, H. Willard, J. Moeschler and C. Hall for sharing resources and their clinical and scientific expertise. This work was supported by NIH grants AR-43827, HD-22657 and HD-01205, and by the March of Dimes Birth Defects Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gong, Y., Krakow, D., Marcelino, J. et al. Heterozygous mutations in the gene encoding noggin affect human joint morphogenesis. Nat Genet 21, 302–304 (1999). https://doi.org/10.1038/6821
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/6821
This article is cited by
-
Noggin promotes osteogenesis in human adipose-derived mesenchymal stem cells via FGFR2/Src/Akt and ERK signaling pathway
Scientific Reports (2024)
-
Identification of a novel nonsense NOG mutation in a patient with stapes ankylosis and symphalangism spectrum disorder
Human Genome Variation (2023)
-
Genetic and clinical phenotypic analysis of familial stapes sclerosis caused by an NOG mutation
BMC Medical Genomics (2020)
-
Identification of a novel mutation of NOG in family with proximal symphalangism and early genetic counseling
BMC Medical Genetics (2019)
-
Novel NOG (p.P42S) mutation causes proximal symphalangism in a four-generation Chinese family
BMC Medical Genetics (2019)