Abstract
Background
We conducted a prospective phase II trial of IrOx in patients with advanced non-small cell lung cancer to evaluate the efficacy and toxicity.
Patients and methods
Patients with histologically or cytologically proven non-small cell lung cancer (NSCLC), aged ≥18 years, Eastern Cooperative Oncology Group performance status 0–1, at stage IIIB (pleural effusion)/IV or with recurrent disease not suitable for primary surgical treatment, with no palliative chemotherapy or radiotherapy to the chest or immunotherapy or biologic therapy, the presence of measurable disease by RECIST, and who had given signed written informed consent, were eligible. Treatment consisted of irinotecan 65 mg/m2 on days 1 and 8 and oxaliplatin 130 mg/m2 on day 1, repeated every 3 weeks.
Results
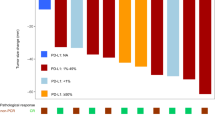
A total of 18 patients were enrolled in June and August 2007, the median age was 59 years (47–73). In total, 71 cycles were administered with a median of 4 cycles per patient (range, 1–6 cycles) and 18 patients were evaluable for treatment response. An independent review of tumor responses gave an overall response rate of 27.7% (CR: 0, PR: 5/18; 95% CI, 7–48.4%) by intent-to-treat analysis. The median overall survival of all patients was 14 months and the median time-to-progression was 4.2 months (95% CI, 1.959–6.441). The most common grade 3/4 toxicities were diarrhea (7% of all cycles) and neutropenia (5.6% of all cycles). Grade 3 peripheral neuropathy occurred in one patient and one patient died due to sepsis.
Conclusion
This study suggests that IrOx combination therapy has moderate activity with a tolerable toxicity profile. However, it was not warranted to evaluate further this regimen as first-line treatment for patients with advanced or metastatic NSCLC using the current dosages and schedule.


Similar content being viewed by others
References
Bae J, Gwack J, Park SK, Shin HR, Chang SH, Yoo KY (2007) Cigarette smoking, alcohol consumption, tuberculosis and risk of lung cancer: the Korean multi-center cancer cohort study. J Prev Med Pub Health 40:321–328
Cappuzzo F, Novello S, De Marinis F, Franciosi V, Maur M, Ceribelli A, Lorusso V, Barbieri F, Castaldini L, Crucitta E, Marini L, Bartolini S, Scagliotti GV, Crino L (2005) Phase II study of gemcitabine plus oxaliplatin as first-line chemotherapy for advanced non-small-cell lung cancer. Br J Cancer 93:29–34
Cardenal F, Domine M, Massuti B, Carrato A, Felip E, Garrido P, Juan O, Artal A, Barneto I, Lopez-Vivanco G, Balcells M, Rosell R (2003) Three-week schedule of irinotecan and cisplatin in advanced non-small cell lung cancer: a multicentre phase II study. Lung Cancer 39:201–207
Chollet P, Bensmaine MA, Brienza S, Deloche C, Cure H, Caillet H, Cvitkovic E (1996) Single agent activity of oxaliplatin in heavily pretreated advanced epithelial ovarian cancer. Ann Oncol 7:1065–1070
Cortinovis D, Bidoli P, Zilembo N, Fusi A, Bajetta E (2008) Oxaliplatin doublets in non-small cell lung cancer: a literature review. Lung Cancer 60:325–331
de Gramont A, Vignoud J, Tournigand C, Louvet C, Andre T, Varette C, Raymond E, Moreau S, Le Bail N, Krulik M (1997) Oxaliplatin with high-dose leucovorin and 5-fluorouracil 48-h continuous infusion in pretreated metastatic colorectal cancer. Eur J Cancer 33:214–219
De Vore RF, Johnson DH, Crawford J, Garst J, Dimery IW, Eckardt J, Eckhardt SG, Elfring GL, Schaaf LJ, Hanover CK, Miller LL (1999) Phase II study of irinotecan plus cisplatin in patients with advanced non-small-cell lung cancer. J Clin Oncol 17:2710–2720
Fukuda M, Oka M, Soda H, Kinoshita A, Nagashima S, Kuba M, Takatani H, Tsurutani J, Nakamura Y, Kasai T, Inoue Y, Soejima Y, Kohno S (2004) Phase II study of irinotecan combined with carboplatin in previously untreated non-small-cell lung cancer. Cancer Chemother Pharmacol 54:573–577
Fukuoka M, Niitani H, Suzuki A, Motomiya M, Hasegawa K, Nishiwaki Y, Kuriyama T, Ariyoshi Y, Negoro S, Masuda N et al (1992) A phase II study of CPT–11, a new derivative of camptothecin, for previously untreated non-small-cell lung cancer. J Clin Oncol 10:16–20
Georgoulias V, Agelidou A, Syrigos K, Rapti A, Agelidou M, Nikolakopoulos J, Polyzos A, Athanasiadis A, Tselepatiotis E, Androulakis N, Kalbakis K, Samonis G, Mavroudis D (2005) Second-line treatment with irinotecan plus cisplatin vs cisplatin of patients with advanced non-small-cell lung cancer pretreated with taxanes and gemcitabine: a multicenter randomised phase II study. Br J Cancer 93:763–769
Goldberg RM, Sargent DJ, Morton RF, Fuchs CS, Ramanathan RK, Williamson SK, Findlay BP, Pitot HC, Alberts SR (2004) A randomized controlled trial of fluorouracil plus leucovorin, irinotecan, and oxaliplatin combinations in patients with previously untreated metastatic colorectal cancer. J Clin Oncol 22:23–30
Guichard S, Arnould S, Hennebelle I, Bugat R, Canal P (2001) Combination of oxaliplatin and irinotecan on human colon cancer cell lines: activity in vitro and in vivo. Anticancer Drugs 12:741–751
Han JY, Lim HS, Shin ES, Yoo YK, Park YH, Lee JE, Jang IJ, Lee DH, Lee JS (2006) Comprehensive analysis of UGT1A polymorphisms predictive for pharmacokinetics and treatment outcome in patients with non-small-cell lung cancer treated with irinotecan and cisplatin. J Clin Oncol 24:2237–2244
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, Thun MJ (2008) Cancer statistics, 2008. CA Cancer J Clin 58:71–96
Kakolyris S, Kouroussis C, Souglakos J, Agelaki S, Kalbakis K, Vardakis N, Vamvakas L, Georgoulias V (2001) Cisplatin and irinotecan (CPT-11) as second-line treatment in patients with advanced non-small cell lung cancer. Lung Cancer 34(Suppl 4):S71–S76
Kim HG, Lee GW, Kang JH, Kang MH, Hwang IG, Kim SH, Hahm JR, Jeong YY, Kim HC, Lee JD, Lee JS, Hwang YS (2008) Combination chemotherapy with irinotecan and cisplatin in elderly patients (≥65 years) with extensive-disease small-cell lung cancer. Lung Cancer 61:220–226
Masuda N, Fukuoka M, Fujita A, Kurita Y, Tsuchiya S, Nagao K, Negoro S, Nishikawa H, Katakami N, Nakagawa K, Niitani H (1998) A phase II trial of combination of CPT-11 and cisplatin for advanced non-small-cell lung cancer. CPT-11 Lung Cancer Study Group. Br J Cancer 78:251–256
McLaren V, Graham J, Paul J, Dunlop D (2008) A phase II study of oxaliplatin and gemcitabine in advanced inoperable stage IIIB/IV non-small cell lung cancer. Clin Oncol (R Coll Radiol) 20:384–385
Monnet I, Brienza S, Hugret F, Voisin S, Gastiaburu J, Saltiel JC, Soulie P, Armand JP, Cvitkovic E, de Cremoux H (1998) Phase II study of oxaliplatin in poor-prognosis non-small cell lung cancer (NSCLC) ATTIT. Association pour le Traitement des Tumeurs Intra Thoraciques. Eur J Cancer 34:1124–1127
Negoro S, Masuda N, Takada Y, Sugiura T, Kudoh S, Katakami N, Ariyoshi Y, Ohashi Y, Niitani H, Fukuoka M (2003) Randomised phase III trial of irinotecan combined with cisplatin for advanced non-small-cell lung cancer. Br J Cancer 88:335–341
Ohe Y, Ohashi Y, Kubota K, Tamura T, Nakagawa K, Negoro S, Nishiwaki Y, Saijo N, Ariyoshi Y, Fukuoka M (2007) Randomized phase III study of cisplatin plus irinotecan versus carboplatin plus paclitaxel, cisplatin plus gemcitabine, and cisplatin plus vinorelbine for advanced non-small-cell lung cancer: Four-Arm Cooperative Study in Japan. Ann Oncol 18:317–323
Pfister DG, Johnson DH, Azzoli CG, Sause W, Smith TJ, Baker S Jr, Olak J, Stover D, Strawn JR, Turrisi AT, Somerfield MR (2004) American Society of Clinical Oncology treatment of unresectable non-small-cell lung cancer guideline: update 2003. J Clin Oncol 22:330–353
Pillot GA, Read WL, Hennenfent KL, Marsh S, Gao F, Viswanathan A, Cummings K, McLeod HL, Govindan R (2006) A phase II study of irinotecan and carboplatin in advanced non-small cell lung cancer with pharmacogenomic analysis: final report. J Thorac Oncol 1:972–978
Raez LE, Santos ES, Lopes G, Rosado MF, Negret LM, Rocha-Lima C, Tolba K, Farfan N, Hamilton-Nelson K, Silva O, Roman E (2006) Efficacy and safety of oxaliplatin and docetaxel in patients with locally advanced and metastatic non-small-cell lung cancer (NSCLC). Lung Cancer 53:347–353
Raymond E, Faivre S, Woynarowski JM, Chaney SG (1998) Oxaliplatin: mechanism of action and antineoplastic activity. Semin Oncol 25:4–12
Scagliotti GV, Kortsik C, Dark GG, Price A, Manegold C, Rosell R, O’Brien M, Peterson PM, Castellano D, Selvaggi G, Novello S, Blatter J, Kayitalire L, Crino L, Paz-Ares L (2005) Pemetrexed combined with oxaliplatin or carboplatin as first-line treatment in advanced non-small cell lung cancer: a multicenter, randomized, phase II trial. Clin Cancer Res 11:690–696
Scagliotti GV, Parikh P, von Pawel J, Biesma B, Vansteenkiste J, Manegold C, Serwatowski P, Gatzemeier U, Digumarti R, Zukin M, Lee JS, Mellemgaard A, Park K, Patil S, Rolski J, Goksel T, de Marinis F, Simms L, Sugarman KP, Gandara D (2008) Phase III study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapy-naive patients with advanced-stage non-small-cell lung cancer. J Clin Oncol 26:3543–3551
Schiller JH, Harrington D, Belani CP, Langer C, Sandler A, Krook J, Zhu J, Johnson DH (2002) Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med 346:92–98
Simon R (1989) Optimal two-stage designs for phase II clinical trials. Control Clin Trials 10:1–10
Takeda K, Takifuji N, Uejima H, Yoshimura N, Terakawa K, Negoro S (2002) Phase II study of irinotecan and carboplatin for advanced non-small cell lung cancer. Lung Cancer 38:303–308
Wakelee HA, Wang W, Schiller JH, Langer CJ, Sandler AB, Belani CP, Johnson DH (2006) Survival differences by sex for patients with advanced non-small cell lung cancer on Eastern Cooperative Oncology Group trial 1594. J Thorac Oncol 1:441–446
Weissman CH, Reynolds CH, Neubauer MA, Steinberg I, Asmar L (2008) Phase III open-label comparison of gemcitabine/oxaliplatin versus paclitaxel/carboplatin as first line therapy for advanced non-small cell lung cancer (NSCLC). J Clin Oncol 26 May 20 suppl; abstr 8024:429s
Wisnivesky JP, Halm EA (2007) Sex differences in lung cancer survival: do tumors behave differently in elderly women? J Clin Oncol 25:1705–1712
Ychou M, Conroy T, Seitz JF, Gourgou S, Hua A, Mery-Mignard D, Kramar A (2003) An open phase I study assessing the feasibility of the triple combination: oxaliplatin plus irinotecan plus leucovorin/5-fluorouracil every 2 weeks in patients with advanced solid tumors. Ann Oncol 14:481–489
Acknowledgments
We acknowledge the Pfizer Pharmaceuticals Korea Ltd. who generously donated irinotecan for the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chang, M.H., Kim, K.H., Jun, H.J. et al. Irinotecan and oxaliplatin combination as the first-line treatment for patients with advanced non-small cell lung cancer. Cancer Chemother Pharmacol 64, 917–924 (2009). https://doi.org/10.1007/s00280-009-0943-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-009-0943-7