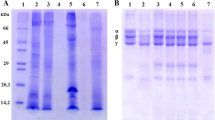
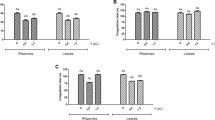
Abstract
Induratia spp. fungi have been poorly evaluated for their non-volatile secondary metabolites. In the present work, we evaluated the effects of non-volatile secondary metabolites released into the culture medium by Induratia spp. upon toxic alterations induced by Bothrops spp. venoms. B. atrox venom phospholipase was inhibited by Induratia spp. around 12 and 16%. The extracts of the two strains inhibited 12–25% of the hemolysis induced by B.moojeni venom. Thrombolysis was inhibited by 30–60% by the compounds present in both extracts. The coagulation induced by B. moojeni venom was prolonged by 26–41 s by the action of the extracts of I. coffeana. The fungal extracts did not exert any cytotoxic effect, nor did they induce any alteration in the other evaluated substrates show the potential use of non-volatile metabolites produced by the fungi evaluated as enzyme modulators, especially for proteases with a fundamental role in human hemostasis.




Similar content being viewed by others
Availability of data and materials
Not applicable.
References
Alurappa R, Chowdappa S, Narayanaswamy R et al (2018) Endophytic fungi and bioactive metabolites production: an update. In: Microbial biotechnology. Springer Singapore, Singapore, pp 455–482
Alves NM, Guimarães LHS, Piccoli RH, Cardoso PG (2016) Production and partial characterization of an extracellular phytase produced by Muscodor sp. under Submerged fermentation. Adv Microbiol 06:23–32. https://doi.org/10.4236/aim.2016.61003
Andrade MA, das Cardoso MG, Preté PSC et al (2018) Toxicological aspects of the essential oil from Cinnamodendron dinisii. Chem Biodivers 15:e1800066. https://doi.org/10.1002/cbdv.201800066
Cardoso MGB, Trento MVC, Reis CH et al (2019) Lecanicillium aphanocladii: snake venom phospholipases A 2 and proteases as tools to prospect enzymatic inhibitors. Lett Appl Microbiol 69:88–95. https://doi.org/10.1111/lam.13171
Cesar PHS, Cardoso Trento MV, Sales TA et al (2019) Molecular interactions between p -coumaric acid and snake venom toxins. J Cell Biochem 120:14594–14603. https://doi.org/10.1002/jcb.28721
Chen J (2019) Confirming the phylogenetic position of the genus Muscodor and the description of a new Muscodor species. Mycosphere 10:187–201. https://doi.org/10.5943/mycosphere/10/1/2
Cintra ACO, De Toni LGB, Sartim MA et al (2012) Batroxase, a new metalloproteinase from B. atrox snake venom with strong fibrinolytic activity. Toxicon 60:70–82. https://doi.org/10.1016/j.toxicon.2012.03.018
da Bastos APSP, Cardoso PG, Santos ÍAFM et al (2020) Enzymatic Modulators from Induratia spp. Curr Microbiol 77:3603–3611. https://doi.org/10.1007/s00284-020-02170-5
Demain AL (2014) Importance of microbial natural products and the need to revitalize their discovery. J Ind Microbiol Biotechnol 41:185–201. https://doi.org/10.1007/s10295-013-1325-z
Guimarães SSC, Tavares DG, Monteiro MCP, Pedroso MP, Nunes CA, Mourão B, Carvalho IS, Bardaji DKR, Camargo ILBC, Lana UGP, Gomes EA, Queiroz MV, Pereira OL, Cardoso PG (2021) Polyphasic characterization and antimicrobial properties of Induratia species from Coffea arabica in Brazil. Mycol Prog. https://doi.org/10.1007/s11557-021-01743-3
Gupta M, Meshram V (2018) The biological promises of endophytic Muscodor Species. In: Fungi and their role in sustainable development: current perspectives. Springer Singapore, Singapore, pp 51–74
Gutiérrez J, Avila C, Rojas E, Cerdas L (1988) An alternative in vitro method for testing the potency of the polyvalent antivenom produced in Costa Rica. Toxicon 26:411–413. https://doi.org/10.1016/0041-0101(88)90010-4
Hongsanan S, Hyde KD, Bahkali AH et al (2015) Fungal biodiversity profiles 11–20. Cryptogam Mycol 36:355–380. https://doi.org/10.7872/crym/v36.iss3.2015.355
Hyde KD, Xu J, Rapior S et al (2019) The amazing potential of fungi: 50 ways we can exploit fungi industrially. Fungal Divers 97:1–136. https://doi.org/10.1007/s13225-019-00430-9
Kaddes A, Fauconnier M-L, Sassi K et al (2019) Endophytic fungal volatile compounds as solution for sustainable agriculture. Molecules 24:1065. https://doi.org/10.3390/molecules24061065
Kapoor N, Saxena S (2016) Xanthine oxidase inhibitory and antioxidant potential of Indian Muscodor species. 3 Biotech 6:248. https://doi.org/10.1007/s13205-016-0569-5
Kaul S, Gupta S, Ahmed M, Dhar MK (2012) Endophytic fungi from medicinal plants: a treasure hunt for bioactive metabolites. Phytochem Rev 11:487–505. https://doi.org/10.1007/s11101-012-9260-6
Kini RM (2011) Toxins in thrombosis and haemostasis: potential beyond imagination. J Thromb Haemost 9:195–208. https://doi.org/10.1111/j.1538-7836.2011.04279.x
Kusari S, Spiteller M (2011) Are we ready for industrial production of bioactive plant secondary metabolites utilizing endophytes? Nat Prod Rep 28:1203. https://doi.org/10.1039/c1np00030f
Lago J, Toledo-Arruda A, Mernak M et al (2014) Structure-activity association of flavonoids in lung diseases. Molecules 19:3570–3595. https://doi.org/10.3390/molecules19033570
Li R, Li J, Zhou Z et al (2018) Antibacterial and antitumor activity of secondary metabolites of endophytic fungi Ty5 from Dendrobium officinale. J Biobased Mater Bioenergy 12:184–193. https://doi.org/10.1166/jbmb.2018.1769
Macías-Rubalcava ML, Hernández-Bautista BE, Oropeza F et al (2010) Allelochemical effects of volatile compounds and organic extracts from Muscodor yucatanensis, a tropical endophytic fungus from Bursera simaruba. J Chem Ecol 36:1122–1131. https://doi.org/10.1007/s10886-010-9848-5
Marcussi S, Stábeli RG, Santos-Filho NA et al (2013) Genotoxic effect of Bothrops snake venoms and isolated toxins on human lymphocyte DNA. Toxicon 65:9–14. https://doi.org/10.1016/j.toxicon.2012.12.020
Marques TR, Cesar PHS, Braga MA et al (2018) Fruit bagasse phytochemicals from Malpighia Emarginata rich in enzymatic inhibitor with modulatory action on hemostatic processes. J Food Sci 83:2840–2849. https://doi.org/10.1111/1750-3841.14330
McCleary RJR, Kini RM (2013) Snake bites and hemostasis/thrombosis. Thromb Res 132:642–646. https://doi.org/10.1016/j.thromres.2013.09.031
Meshram V, Saxena S, Kapoor N (2014) Muscodor strobelii, a new endophytic species from South India. Mycotaxon 128:93–104. https://doi.org/10.5248/128.93
Monteiro MCP, Alves NM, de Queiroz MV et al (2017) Antimicrobial activity of endophytic fungi from coffee plants. Biosci J. https://doi.org/10.14393/BJ-v33n2-34494
Monteiro MCP, Tavares DG, Nery EM et al (2020) Enzyme production by Induratia spp. isolated from coffee plants in Brazil. Braz Arch Biol Technol. https://doi.org/10.1590/1678-4324-2020180673
Mota SF, Pádua PF, Ferreira AN, Gomes LBW, Dias MA, Souza EA, Pereira OL, Cardoso PG (2021) Biological control of common bean diseases using endophytic Induratia spp. Biol Control 159(2):104629. https://doi.org/10.1016/j.biocontrol.2021.104629
Oliveira CHM, Simão AA, Trento MVC et al (2016) Inhibition of proteases and phospholipases A2 from Bothrops atrox and Crotalus durissus terrificus snake venoms by ascorbic acid, vitamin E, and B-complex vitamins. An Acad Bras Cienc 88:2005–2016. https://doi.org/10.1590/0001-3765201620160030
Pena LC, Jung LF, Savi DC et al (2017) A Muscodor strain isolated from Citrus sinensis and its production of volatile organic compounds inhibiting Phyllosticta citricarpa growth. J Plant Dis Prot 124:349–360. https://doi.org/10.1007/s41348-016-0065-5
Qadri M, Nalli Y, Jain SK et al (2017) An insight into the secondary metabolism of Muscodor yucatanensis: small-molecule epigenetic modifiers induce expression of secondary metabolism-related genes and production of new metabolites in the endophyte. Microb Ecol 73:954–965. https://doi.org/10.1007/s00248-016-0901-y
Ratnasooriya WD, Jayakody JRAC, Premakumara GAS, Ediriweera ERHSS (2005) Antioxidant activity of water extract of Scoparia dulcis. Fitoterapia 76:220–222. https://doi.org/10.1016/j.fitote.2004.06.012
Rodrigues VM, Soares AM, Guerra-Sá R et al (2000) Structural and functional characterization of neuwiedase, a nonhemorrhagic fibrin(ogen)olytic metalloprotease from Bothrops neuwiedi snake venom. Arch Biochem Biophys 381:213–224. https://doi.org/10.1006/abbi.2000.1958
Samarakoon MC, Thongbai B, Hyde KD et al (2020) Elucidation of the life cycle of the endophytic genus Muscodor and its transfer to Induratia in Induratiaceae fam. nov., based on a polyphasic taxonomic approach. Fungal Divers 101:177–210. https://doi.org/10.1007/s13225-020-00443-9
Saxena S, Meshram V, Kapoor N (2015) Muscodor tigerii sp. nov.-Volatile antibiotic producing endophytic fungus from the Northeastern Himalayas. Ann Microbiol 65:47–57. https://doi.org/10.1007/s13213-014-0834-y
Sears J, Dirkse E, Markworth C, Strobel GA (2001) Volatile antimicrobials from Muscodor albus, a novel endophytic fungus. Microbiology 147:2943–2950. https://doi.org/10.1099/00221287-147-11-2943
Shipunov A, Newcombe G, Raghavendra AKH, Anderson CL (2008) Hidden diversity of endophytic fungi in an invasive plant. Am J Bot 95:1096–1108. https://doi.org/10.3732/ajb.0800024
Strobel G (2011) Muscodor species- endophytes with biological promise. Phytochem Rev 10:165–172. https://doi.org/10.1007/s11101-010-9163-3
Strobel GA (2014) Methods of discovery and techniques to study endophytic fungi producing fuel-related hydrocarbons. Nat Prod Rep 31:259. https://doi.org/10.1039/c3np70129h
Suwannarach N, Kumla J, Bussaban B et al (2013) Molecular and morphological evidence support four new species in the genus Muscodor from northern Thailand. Ann Microbiol 63:1341–1351. https://doi.org/10.1007/s13213-012-0593-6
Zhang C-L, Wang G-P, Mao L-J et al (2010) Muscodor fengyangensis sp. nov. from southeast China: morphology, physiology and production of volatile compounds. Fungal Biol 114:797–808. https://doi.org/10.1016/j.funbio.2010.07.006
Acknowledgements
The authors express their gratitude to the Universidade Federal de Lavras for the structural and logistical support. We also acknowledge the support of the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Nível Superior (CAPES), and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) which made this project possible.
Funding
Conselho Nacional de Desenvolvimento Científico e Tecnológico, Coordenação de Aperfeiçoamento de Nível Superior, and Fundação de Amparo à Pesquisa do Estado de Minas Gerais.
Author information
Authors and Affiliations
Contributions
MGBC: conceptualization, methodology, validation, formal analysis, investigation, data curation, and writing. MVCT: writing, review, and editing. PHSC: writing, review, editing, and creation of images. SM: conceptualization, methodology, resources, writing, review, editing, and project administration. PGC: conceptualization, resources, writing, review, editing, supervision, and funding acquisition. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest, financial or otherwise.
Ethical approval and consent to participate
The methods that required the use of human biological material (cells and blood components) were previously approved by UFLA’s Human Research Ethics Committee (COEP), under the registration number CAAE/80767417.0.0000.5148.
Human and animal rights
No humans were used in this research. All research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2008 (http://www.wma.net/en/20activities/10ethics/10helsinki/).
Consent for publication
An informed consent was taken from all individuals.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cardoso, M.G.B., Trento, M.V.C., Cesar, P.H.S. et al. Metabolites from Induratia spp. modulating key enzymes in human hemostasis. Arch Microbiol 204, 27 (2022). https://doi.org/10.1007/s00203-021-02682-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-021-02682-z